Prophylactic Mitomycin-C Application for Haze Prevention after Photorefractive Keratectomy in Myopia: A Systematic Review
February 12, 2024
Is it Safe for Pregnant Women to Eat Imitation Crab? What You Need to Know
February 18, 2024Understanding Palbociclib (Ibrance) in Advanced Breast Cancer Treatment

Photo by Rebekah Vos on Unsplash
Take away points
- Palbociclib (Ibrance) is a CDK4/6 inhibitor used in combination with aromatase inhibitors or fulvestrant for hormone receptor-positive (HR+) human epidermal growth factor receptor 2 (HER2)-negative advanced/metastatic breast cancer (ABC/MBC).
- Real-world studies in Canada, Argentina, and the US have shown that palbociclib combinations are well tolerated and effective, with low dose reduction rates and high progression-free and survival rates.
- Palbociclib in combination with endocrine therapy has demonstrated improved progression-free survival (PFS) and overall survival (OS) in patients with HR+/HER2- metastatic breast cancer compared to endocrine therapy alone.
- The FDA has approved palbociclib for use in combination with fulvestrant for the treatment of women with HR-positive, HER2-negative advanced or metastatic breast cancer with disease progression following endocrine therapy.
- Genomic complexity detected by noninvasive profiling of blood tumor mutational burden (bTMB) and blood copy-number burden (bCNB) can predict poor outcomes in patients treated with endocrine therapy and CDK4/6 inhibitors (CDK4/6i).
- The addition of palbociclib to endocrine therapy significantly enhances overall survival of HR+/HER2- metastatic breast cancer patients compared to endocrine therapy alone in the second-line setting.
- Non-luminal subtype and high levels of CCNE1 are associated with resistance to CDK4/6 inhibitors like Ibrance in HR+ MBC, and PLK1 inhibition may reverse resistance to palbociclib+ET. Always remember to tell your doctor about any side effects during treatment.
- The GEICAM/2006–10 study did not show a statistically significant increase in disease-free survival by adding adjuvant fulvestrant to anastrozole for postmenopausal women with HR+/HER2- early breast cancer.
Fast references
- Real-World Palbociclib Use in HR+/HER2− Advanced Breast Cancer in Canada
- Palbociclib (Ibrance®) in combination with fulvestrant for the treatment of hormone-receptor-positive, HER2-negative advanced or metastatic breast cancer
- Real-World Tumor Response of Palbociclib in Combination With an Aromatase Inhibitor as First-Line Therapy in Pre/Perimenopausal Women With Metastatic Breast Cancer
- The effects of adding palbociclib to endocrine therapy to treat advanced breast cancer: a plain language summary of a study using the PALOMA-2 and PALOMA-3 trial results.
- FDA Approval of Palbociclib in Combination with Fulvestrant for the Treatment of Hormone Receptor–Positive, HER2-Negative Metastatic Breast Cancer
- Palbociclib: A Review in HR-Positive, HER2-Negative, Advanced or Metastatic Breast Cancer
When it comes to dealing with advanced breast cancer, understanding the available treatment options and their mechanisms is crucial. Palbociclib, commonly known as Ibrance, has emerged as a significant development in the field of cancer treatment, particularly for patients with metastatic breast cancer. This article aims to shed light on the role and significance of palbociclib in the context of advanced breast cancer therapy.
Overview of Palbociclib

What is Palbociclib?
Palbociclib is an inhibitor, specifically a cyclin-dependent kinase 4/6 (CDK 4/6) inhibitor. It is commonly used in combination with hormone therapies, such as letrozole or fulvestrant, for the treatment of hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced or metastatic breast cancer. The action of Palbociclib, a CDK4 inhibitor, involves targeting and halting the CDK 4/6 receptors in cancer cells, thereby decelerating the disease progression.
The Role of Palbociclib in Breast Cancer Treatment
Ibrance is used and plays a vital role in the targeted therapy for breast cancer, particularly in cases where the cancer has spread to other parts of the body. It has shown promising results in clinical trials, demonstrating its efficacy in slowing down the growth and spread of metastatic breast cancer. When used in combination with hormone therapies, palbociclib has become an integral part of the treatment regimen for patients with advanced breast cancer, offering a new hope in managing the disease.
Understanding Advanced Breast Cancer

Advanced breast cancer refers to breast cancer that has spread beyond the breast and nearby lymph nodes to other parts of the body, such as the bones, liver, or lungs. It is also known as metastatic breast cancer and is considered an advanced stage of the disease. At this stage, the cancer cells have the potential to invade and affect distant organs, making treatment more challenging.
Definition of Advanced Breast Cancer
Advanced breast cancer, also known as stage IV or metastatic breast cancer, is defined by the spread of cancer cells from the breast to other parts of the body, such as the bones, liver, lungs, or brain. This stage of breast cancer presents significant treatment challenges due to its ability to affect multiple organs and systems within the body. Patients with advanced breast cancer require comprehensive and targeted treatment approaches, like treatment with palbociclib, to manage the disease and improve their quality of life. It’s important that the doctor may constantly monitor the process for any serious side effects.
Treatment Options for Advanced Breast Cancer
Treatment for advanced breast cancer may include a combination of systemic therapies, such as chemotherapy, hormone therapy, targeted therapy, and immunotherapy, depending on the specific characteristics of the cancer and the individual patient’s health status. Palliative care and supportive therapies also play a crucial role in managing symptoms and improving the overall well-being of patients with advanced breast cancer.
Effectiveness of Palbociclib in Metastatic Breast Cancer
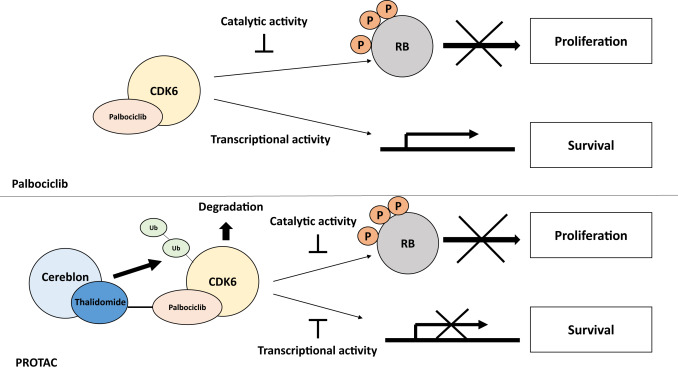
Palbociclib, also known by the brand name Ibrance, has demonstrated significant effectiveness in the treatment of metastatic breast cancer, particularly in hormone receptor-positive, human epidermal growth factor receptor 2-negative cases. Clinical studies and trials have shown promising results regarding the efficacy of palbociclib in slowing down disease progression and improving patient outcomes.
Clinical Trials and Studies on Palbociclib
Clinical trials and research studies have evaluated the effectiveness of palbociclib, both as a monotherapy and in combination with other treatments, over a course of 21 days, in patients with metastatic breast cancer. If any serious side effects are noticed, it’s important to tell your doctor immediately. These studies have provided valuable insights into the role of palbociclib in targeting specific molecular pathways involved in cancer cell proliferation and progression, thereby contributing to advancements in the management of advanced breast cancer.
Using Palbociclib in Metastatic Breast Cancer
Palbociclib is commonly used in combination with hormone therapies, such as letrozole or fulvestrant, for the treatment of hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced or metastatic breast cancer. Its mechanism of action as a cyclin-dependent kinase 4/6 (CDK 4/6) inhibitor has provided a targeted approach to slowing down the growth and spread of metastatic breast cancer, offering new hope to patients in managing the disease.
Managing Side Effects of Palbociclib

Understanding the potential side effects of palbociclib (Ibrance) treatment is essential for patients and healthcare providers to facilitate proactive management strategies. Common side effects experienced by patients taking palbociclib may include fatigue, nausea, diarrhea, and decreased appetite. While these side effects are manageable, they can impact the quality of life and treatment adherence.
Common Side Effects of Palbociclib
It is important for patients to communicate openly with their healthcare team about any side effects they experience while taking palbociclib. Fatigue, a prevalent side effect, can significantly affect daily activities and overall well-being. Nausea and diarrhea may occur in some patients, leading to discomfort and potential nutritional challenges. Additionally, decreased appetite can contribute to nutritional deficiencies and reduced energy levels, impacting the overall management of the disease which is used to treat with 125 mg Ibrance capsules.
Tips for Managing Side Effects
Patient education on managing common side effects is crucial to empower individuals during their treatment journey. Encouraging adequate hydration, balanced nutrition, and feasible physical activity can aid in mitigating fatigue. Furthermore, dietary modifications, anti-nausea medications, and small, frequent meals can help manage gastrointestinal symptoms. Lastly, patients should be encouraged to discuss any changes in appetite or dietary patterns with their healthcare team to address potential nutritional concerns.
Important Considerations for Patients Taking Ibrance
Information Point
Photo by Waldemar on Unsplash
Patients prescribed with palbociclib (Ibrance) should be aware of important considerations related to its use and potential impact on their overall health. Understanding the implications of palbociclib treatment on secondary breast cancer and receiving essential information from healthcare professionals are vital for informed decision-making.
Important Information for Patients Taking Palbociclib
Patients should be informed about the necessity of regular monitoring and follow-up appointments while taking palbociclib to assess treatment response and manage potential side effects. Additionally, patients should receive guidance on medication adherence and the importance of reporting any adverse reactions promptly to their healthcare provider.
Effects of Palbociclib on Secondary Breast Cancer
Patients with secondary breast cancer should be informed about the specific impact of palbociclib on their condition. It is essential for healthcare providers to educate patients about the potential benefits and risks associated with palbociclib treatment, considering the unique characteristics of secondary breast cancer. Patients’ understanding of the treatment’s impact on their health empowers them to actively participate in their care and make informed decisions alongside their healthcare team.
Full references
- A. Llombart-Cussac et al. “Second-line endocrine therapy (ET) with or without palbociclib (P) maintenance in patients (pts) with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2[-]) advanced breast cancer (ABC): PALMIRA trial.” Journal of Clinical Oncology, June 1, 2023. PDF | Semanticscholar
- C. Isaacs et al. “Efficacy and safety of palbociclib plus endocrine therapy in Black and Hispanic patients with hormone receptor positive/human epidermal growth factor receptor 2-negative advanced breast cancer (HR+/HER2- ABC) participating in the PALOMA trials.” Cancer Research, February 15, 2022. PDF | Semanticscholar
- M. Sawaki et al. “Real-world treatment duration of subsequent therapy after palbociclib (PAL) in patients with hormone receptor-positive (HR+)/human epidermal growth factor receptor 2-negative (HER2−) advanced breast cancer (ABC) in Japan.” Cancer Research, March 1, 2023. PDF | Semanticscholar
- P. Bedard et al. “Long-term safety of inavolisib (GDC-0077) in an ongoing phase 1/1b study evaluating monotherapy and in combination (combo) with palbociclib and/or endocrine therapy in patients (pts) with PIK3CA-mutated, hormone receptor-positive/HER2-negative (HR+/HER2-) metastatic breast cancer (BC).” Journal of Clinical Oncology, June 1, 2022. PDF | Semanticscholar
- C. O’Sullivan et al. “Serum thymidine kinase 1 activity (TKa) levels and progression-free survival (PFS) in patients (pts) with hormone receptor positive (HR+) HER2-negative metastatic breast cancer (MBC) on palbociclib (Pb) and endocrine therapy (ET).” Cancer Research, February 15, 2022. PDF | Semanticscholar
- A. Davis et al. “Genomic Complexity Predicts Resistance to Endocrine Therapy and CDK4/6 Inhibition in Hormone Receptor–Positive (HR+)/HER2-Negative Metastatic Breast Cancer.” Clinical Cancer Research, January 24, 2023. PDF | Semanticscholar
- N. Lorman-Carbó et al. “Dissecting the biological activity of different CDK4/6 inhibitors (CDK4/6i) in hormone receptor-positive/HER2-negative (HR+/HER2-) breast cancer (BC).” Cancer Research, March 1, 2023. PDF | Semanticscholar
- Andrea Maliachi Díaz et al. “Comparing efficacy and toxicity in HR+/HER2- metastatic breast cancer patients treated with ribociclib or palbociclib in combination with endocrine therapy: results from a single center in Mexico.” The Breast, October 1, 2023. PDF | Semanticscholar
- M. Ha et al. “Palbociclib plus endocrine therapy significantly enhances overall survival of HR+/HER2− metastatic breast cancer patients compared to endocrine therapy alone in the second‐line setting: A large institutional study.” International Journal of Cancer, February 8, 2022. PDF | Semanticscholar
- H. Rugo et al. “Real-world study of overall survival with palbociclib plus aromatase inhibitor in HR+/HER2− metastatic breast cancer.” NPJ Breast Cancer, October 11, 2022. PDF | Semanticscholar
FAQs
Q: What is Palbociclib (Ibrance) and how is it used in the treatment for breast cancer?
A: Palbociclib, also known as Ibrance, is a drug that is taken by mouth and used mainly in the treatment of breast cancer that is hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-). It also may be taken with food and works by blocking certain proteins in cancer cells, impeding the ability of these cells to grow and divide.
Q: When should someone start the treatment for breast cancer with Palbociclib (Ibrance)?
A: It’s best to talk to your doctor before starting any treatment, including Ibrance. They will consider individual conditions such as the type of breast cancer diagnosed, the progress of the disease, and the patient’s general health to decide if Ibrance is the best choice.
Q: Can Palbociclib (Ibrance) cause side effects?
A: Yes, like many medications, Ibrance may cause side effects. These might include fatigue, nausea, hair thinning, diarrhoea, vomiting, loss of appetite or mouth sores. Some people may experience a decrease in their white blood cells. Your doctor or pharmacist can help you learn about side effects and how to manage them.
Q: How should I manage new or worsening side effects while on Ibrance treatment for breast cancer?
A: If you notice new or worsening side effects while taking Ibrance, it’s important to talk to your doctor or pharmacist as soon as possible. They can provide advice about ways to manage these side effects and adjust the treatment program if necessary.
Q: How often do I have to take Palbociclib (Ibrance) as part of my treatment for breast cancer?
A: Ibrance is commonly taken once a day for 21 days straight, followed by a 7-day break. This is generally referred to as a 28-day cycle. However, the specific direction of how you should take it will be given by your doctor.
Q: What is the importance of monitoring the white blood cells during Ibrance treatment for breast cancer?
A: Monitoring your white blood cell count is important because Ibrance can lower these cell counts, leading to an increased risk of infections. For this reason, blood cell counts should be tested before starting treatment and for at least the first six cycles of therapy.
Q: Is Palbociclib (Ibrance) effective for all types of breast cancer?
A: Ibrance is especially beneficial in treating certain types of breast cancer such as HR+ and HER2- breast cancer. It is also used in combination with the drug fulvestrant for those whose breast cancer progressed after hormonal therapy.
Q: What should patients know about swallowing Ibrance capsules?
A: Ibrance is a medication you take by mouth. The capsules should not be chewed, opened, crushed or split. If you have difficulty swallowing them, talk to your doctor or pharmacist for advice.