The Year You Think You Had — And the One You Actually Lived
April 23, 2026
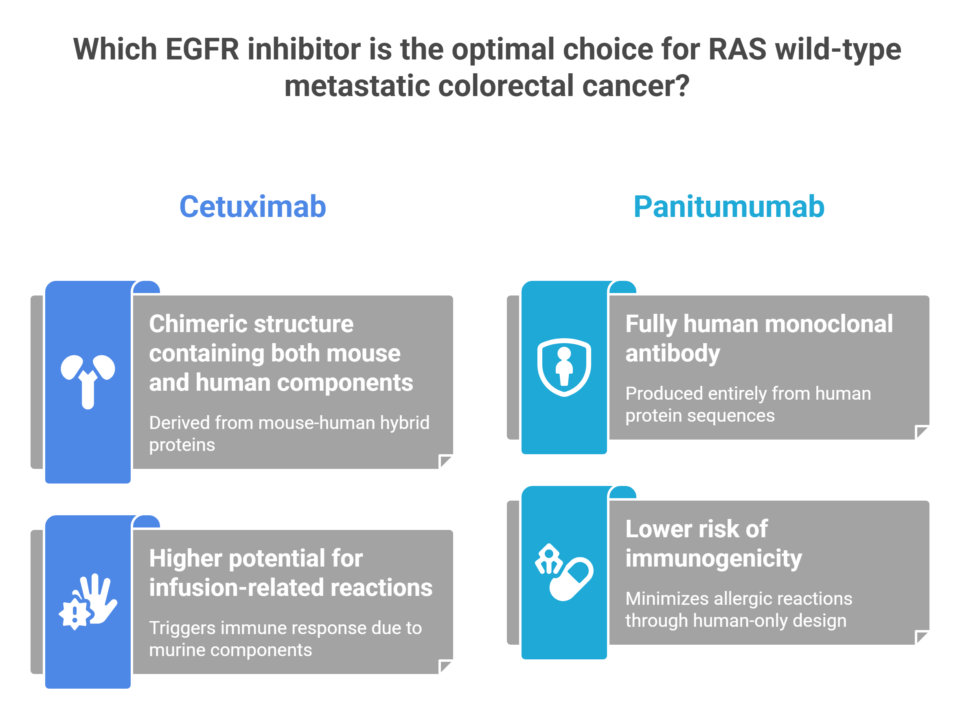
Two Keys, One Lock: The Real Difference Between Cetuximab and Panitumumab
May 1, 2026There is a label on some of the most expensive, most sophisticated medicines ever manufactured. It doesn’t describe the drug’s mechanism. It doesn’t warn about overdose. It says, simply: Do Not Shake. To anyone outside the pharmacy clean room, that instruction sounds faintly absurd — the kind of overcaution printed on products by lawyers afraid of freak accidents. But that label is the last line of defense between a patient and a catastrophic infusion reaction. And the reason it exists reaches all the way down to the sub-molecular architecture of proteins that behave, under stress, less like a drug and more like a crumpling skyscraper.
Monoclonal antibodies — the class of biologics at the center of modern cancer therapy and immunology — are among the most structurally complex molecules ever used as medicine. Understanding why they break, and what breaking costs, is one of the most important stories in contemporary pharmacology.
A Skyscraper Made of Folds
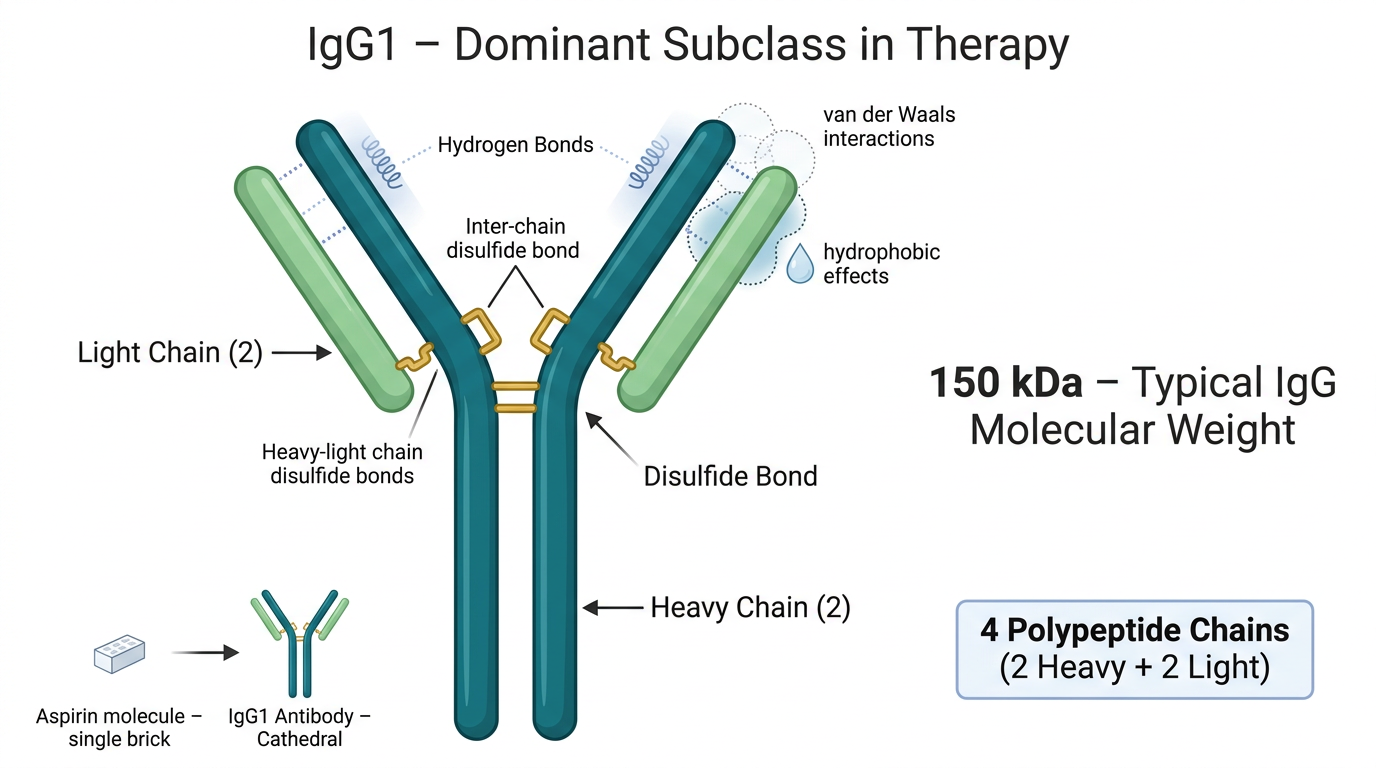
To appreciate the stakes, you have to start with the structure. According to the structure of monoclonal antibodies documented by NCBI, a standard IgG antibody weighs approximately 150 kilodaltons — roughly 150,000 times the mass of a single hydrogen atom. That number alone tells you something. A small-molecule drug like aspirin is a single brick. A monoclonal antibody is a cathedral.
Each IgG molecule is built from two heavy chains and two light chains — a quartet of polypeptides held together by disulfide bonds, as described in Janeway’s Immunobiology via NCBI Bookshelf. The two heavy chains link to each other; each heavy chain links to a light chain. The entire assembly folds into a precise three-dimensional shape maintained not by rigid chemical bonds, but by softer, more easily disturbed forces — hydrogen bonds, van der Waals interactions, hydrophobic effects.
This is the vulnerability. When you shake a vial, you introduce kinetic energy into a system designed to be still. You create air bubbles, and with them, air-liquid interfaces — surfaces where the protein molecules migrate, unfold, and expose the hydrophobic regions normally buried in their interiors. These exposed regions do what hydrophobic surfaces always do in water: they find each other. They clump. They aggregate. And aggregated proteins, unlike their folded counterparts, cannot bind to their targets. The cathedral has collapsed into rubble.
Rituximab: The Canary in the Clean Room
No drug has become more synonymous with the “Do Not Shake” rule than rituximab. It is, by historical measure, a landmark molecule. As detailed in the pharmacology and properties of rituximab via StatPearls, it was the first monoclonal antibody approved for therapeutic use, receiving FDA clearance in 1997. It is an anti-CD20 chimeric antibody — meaning it combines murine variable regions with human IgG1 constant regions — and that IgG1 backbone is not incidental. As documented across multiple reviews of therapeutic mAbs, currently marketed monoclonal antibodies are predominantly of the IgG1 subclass, with IgG2 and IgG4 playing lesser roles.
Why Aggregation Isn’t Just a Potency Problem
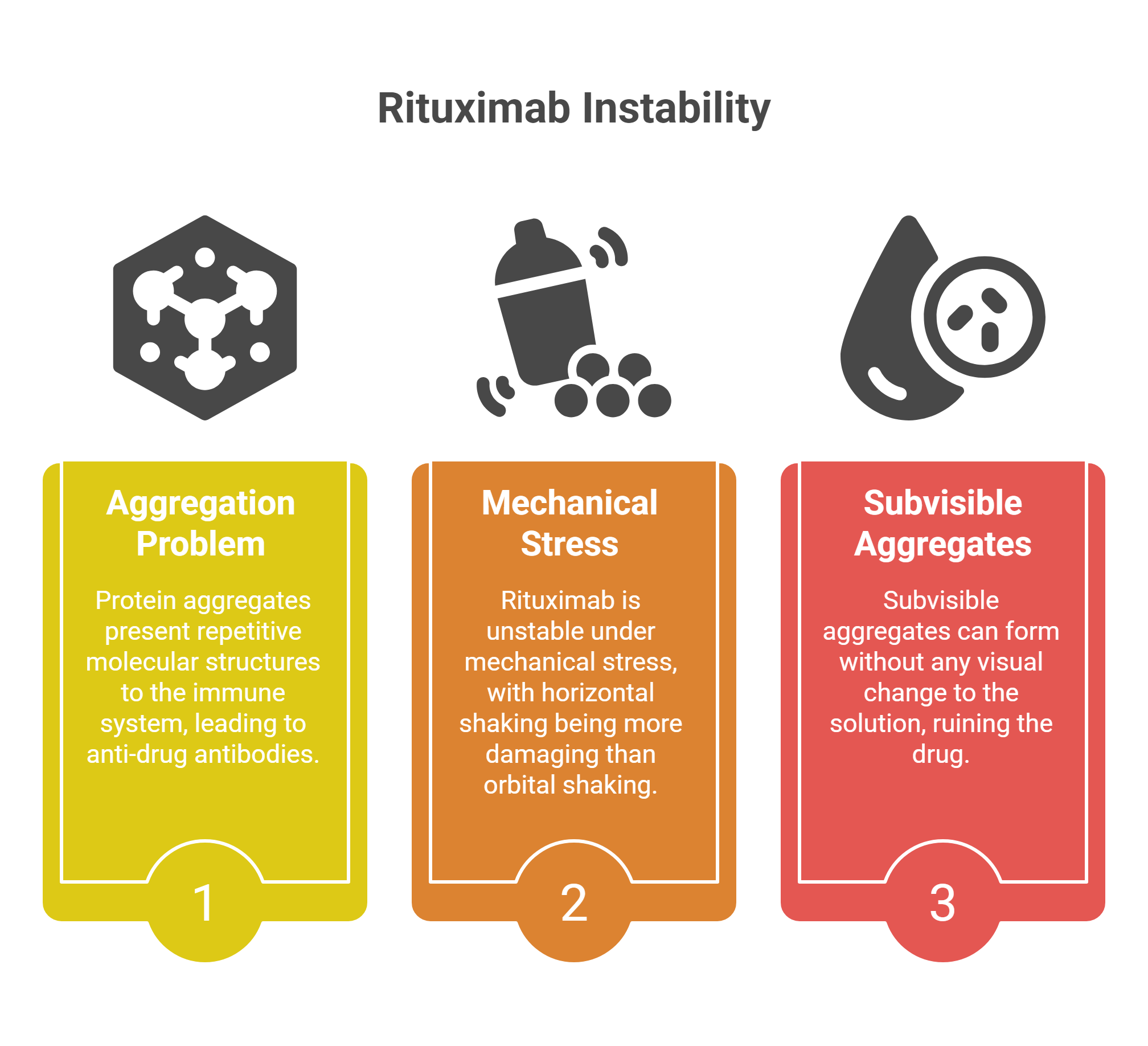
When protein aggregates form, they don’t simply stop working. They present repetitive molecular structures to the immune system — structures that look, to a patrolling B-cell, like the surface of a pathogen. The immune system responds by generating anti-drug antibodies (ADAs). Some ADAs are neutralizing, physically blocking the drug from acting. Others accelerate the drug’s clearance from the body. Either way, the patient isn’t receiving the treatment prescribed.
The instability of rituximab under mechanical stress is well-documented. Even the type of mixing matters: research has shown that horizontal shaking — the back-and-forth motion of a tray sliding across a counter — is far more damaging than orbital shaking, because it creates more violent interfacial movement and what researchers call “film rupture” at the protein surface. Subvisible aggregates can form without any visual change to the solution. The bag looks clear. The drug is ruined.
When Biology Betrayed the Trial: TGN1412
To understand what immune dysregulation from a mishandled biologic can look like at its most extreme, consider what happened in a London clinic in March 2006. Six healthy male volunteers received a single intravenous dose of TGN1412 — a novel superagonist anti-CD28 monoclonal antibody that directly stimulates T cells — as part of a phase I clinical trial. Within 90 minutes, as documented in the landmark NEJM paper by Suntharalingam et al., all six volunteers had developed a systemic inflammatory response: headache, myalgias, nausea, erythema, vasodilatation, and hypotension. Two required intensive organ support for eight and sixteen days respectively.
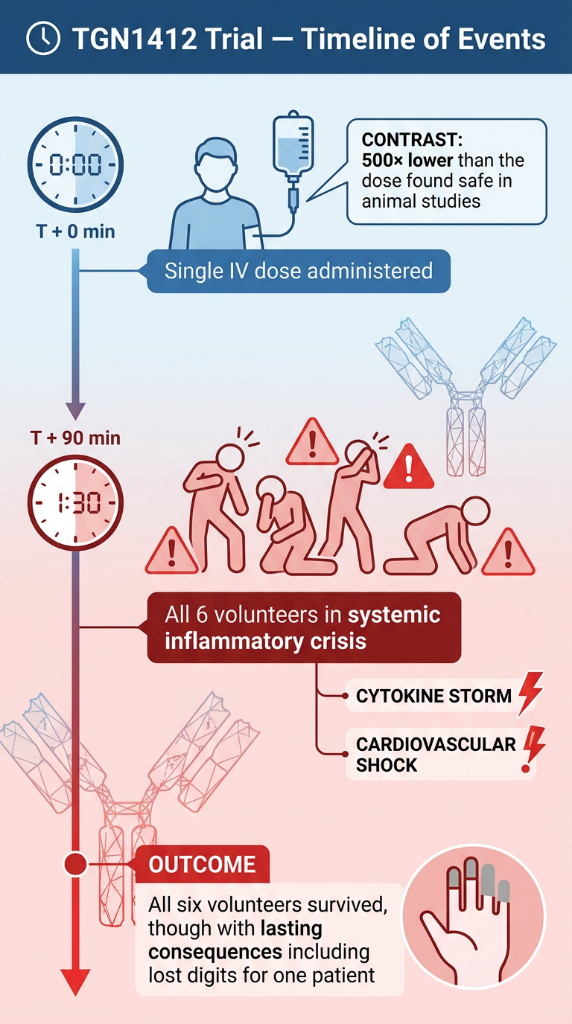
The cause, as established by the MHRA and confirmed in the subsequent scientific analysis “TGN1412: From Discovery to Disaster”, was not a manufacturing error or a dosing mistake. It was unpredicted biological superagonism — the drug behaved in humans in a way that animal models had entirely failed to anticipate. The dose was not too high. It was, in fact, 500 times lower than the dose found safe in preclinical trials. The lesson is not simply that biologics are powerful. It is that their interaction with the human immune system operates on terms we are still learning to read.
The Three Words That Protect Everything
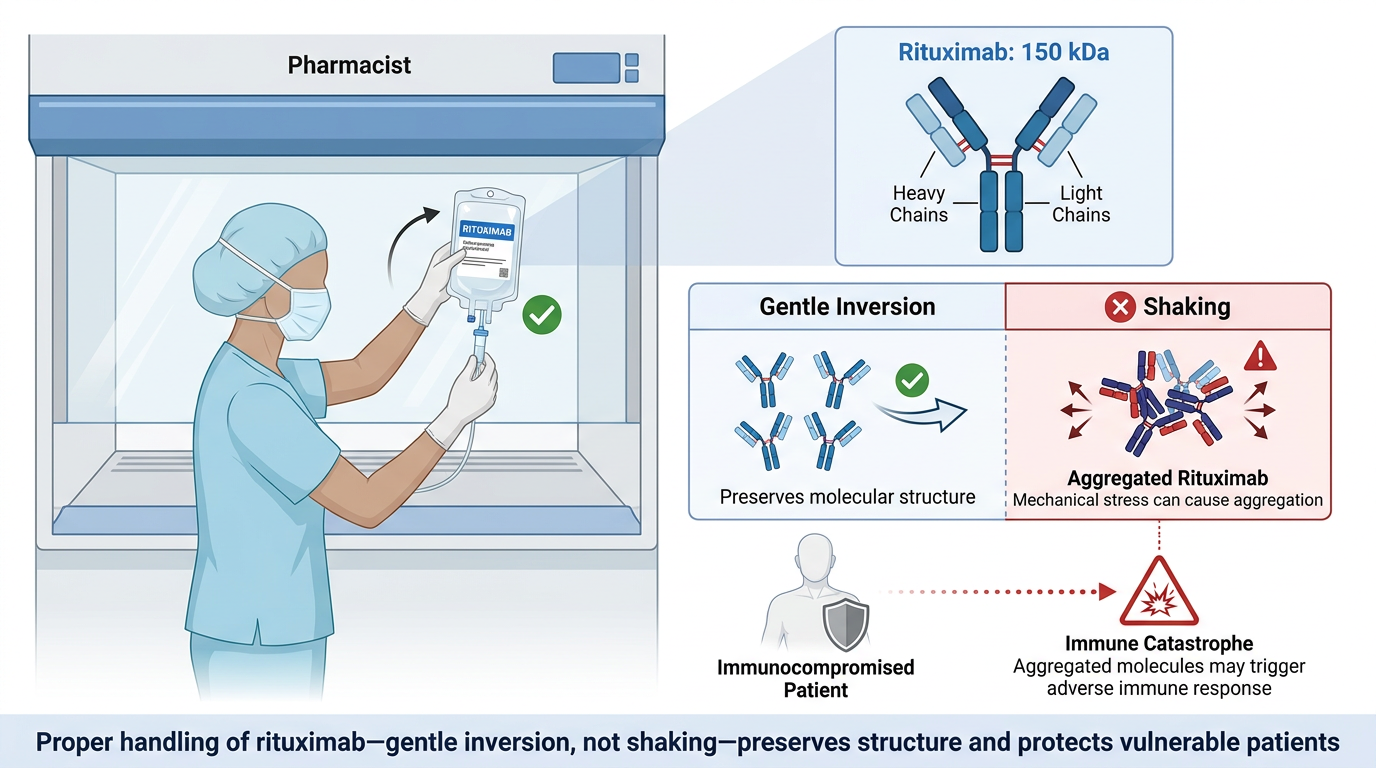
The pharmacist standing at the hood, inverting a bag of rituximab instead of shaking it, is not following an arbitrary ritual. They are honoring the reality of what that molecule is: a 150-kilodalton architecture of two heavy chains and two light chains, held in shape by forces that mechanical stress can undo in seconds. They are protecting a patient whose immune system is already under siege, from a drug that — if aggregated, if mishandled, if delivered in a form the body reads as hostile — could trigger the very immunological catastrophe it was designed to prevent.
“Do Not Shake” is three words. Behind them is the entire history of protein chemistry, two decades of hard-won clinical pharmacology, and the lesson of six men who nearly died in a London clinic on a Tuesday morning in 2006. The label is not overcaution. It is a summary of everything we know about what happens when we stop treating these molecules with the gravity they deserve.
Verified Sources
- NCBI Bookshelf — Immunobiology (Janeway et al.) · IgG structure and molecular weight
- PMC / NIH — Pharmacokinetics of Monoclonal Antibodies · IgG1 prevalence in therapeutics
- StatPearls / NCBI — Rituximab: pharmacology, history, and IgG1 classification
- PubMed / NEJM — Suntharalingam et al., 2006 · TGN1412 Phase I Trial
- PMC / NIH — “TGN1412: From Discovery to Disaster”