Defending the Skeleton After CDK4/6 Inhibition
April 21, 2026
The Year You Think You Had — And the One You Actually Lived
April 23, 2026Every year, roughly 275,000 cancer patients in the United States receive 5-fluorouracil. It is one of oncology’s most enduring weapons — and one of its most dangerous. More than 1,300 patients die each year from 5-FU–related toxicity alone. The difference between the drug that saves a life and the drug that ends one often comes down to a molecule most people would mistake for a vitamin: leucovorin.
The Superglue That Changes Everything
Source: Meta-analysis of 9 randomized controlled trials in advanced colorectal cancer
When leucovorin is paired with methotrexate, it acts as a rescue agent, shielding healthy cells from collateral damage. But pair it with 5-fluorouracil, and its role inverts completely. It becomes a biochemical potentiator — a molecule that makes the chemotherapy more lethal.
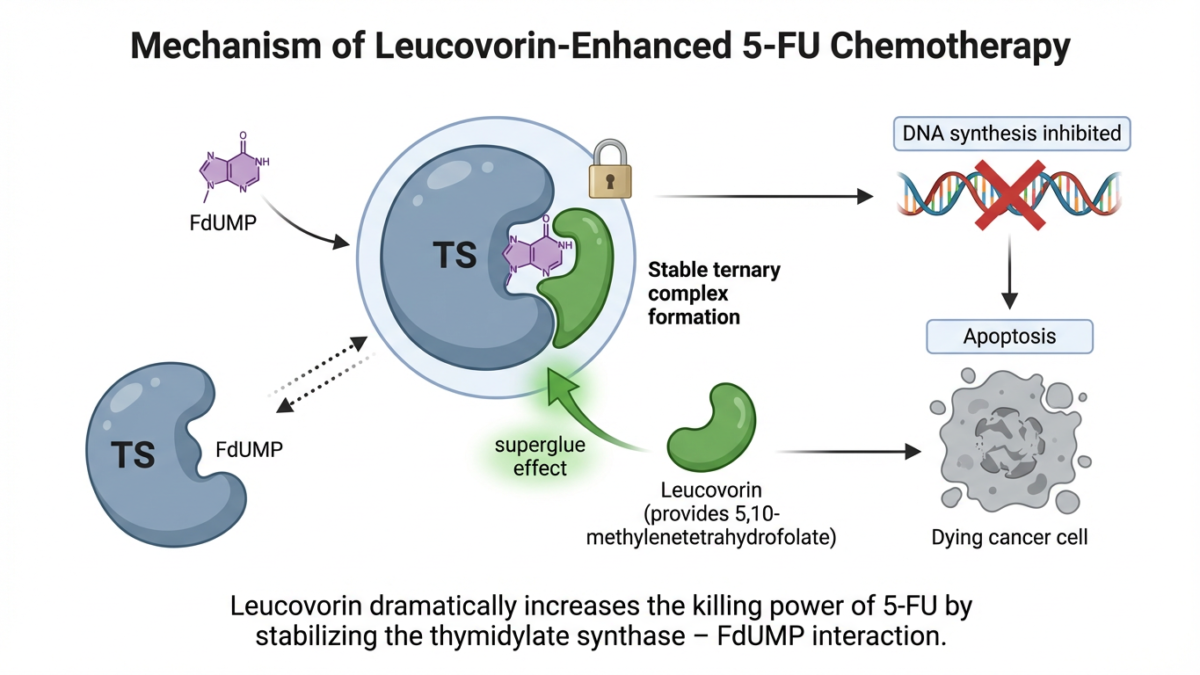
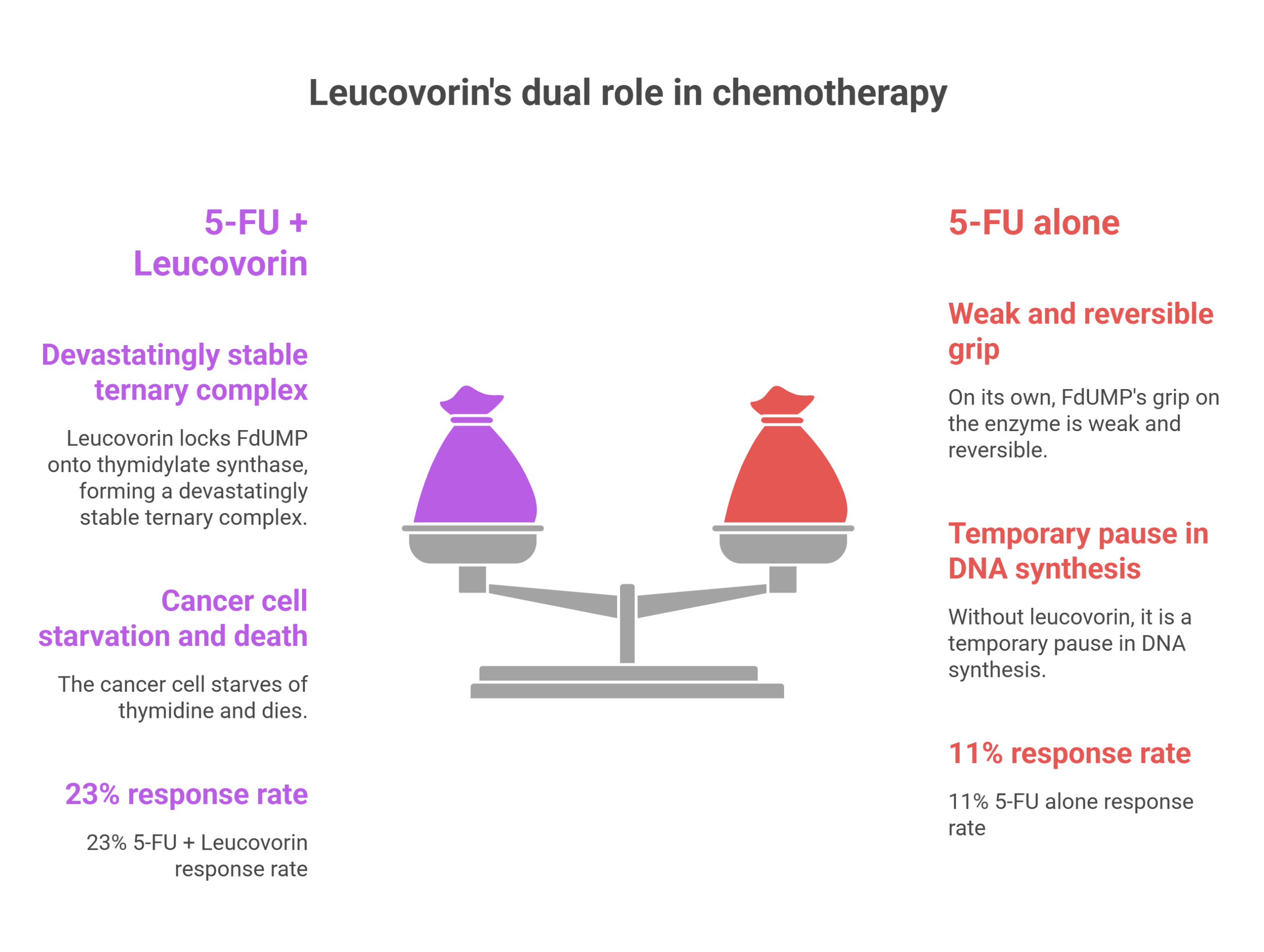
The reason lies in a molecular structure called the ternary complex. 5-FU is metabolized inside cells into FdUMP, a decoy that mimics a natural building block of DNA. FdUMP tricks the enzyme thymidylate synthase — the cellular machinery responsible for manufacturing thymidine, an essential ingredient for DNA replication — and jams it. But on its own, FdUMP’s grip on the enzyme is weak and reversible. The drug eventually drifts away, and the enzyme resumes work.
Leucovorin changes that equation. As a reduced folate, it bypasses DHFR and enters the folate pool directly, flooding cells with 5,10-methylenetetrahydrofolate. That cofactor acts as molecular superglue, locking FdUMP onto thymidylate synthase and forming a devastatingly stable ternary complex of enzyme, drug, and folate. Without leucovorin, it is a temporary pause in DNA synthesis. With it, the cancer cell starves of thymidine and dies.
A Three-Pronged Attack — and Its Gatekeeper
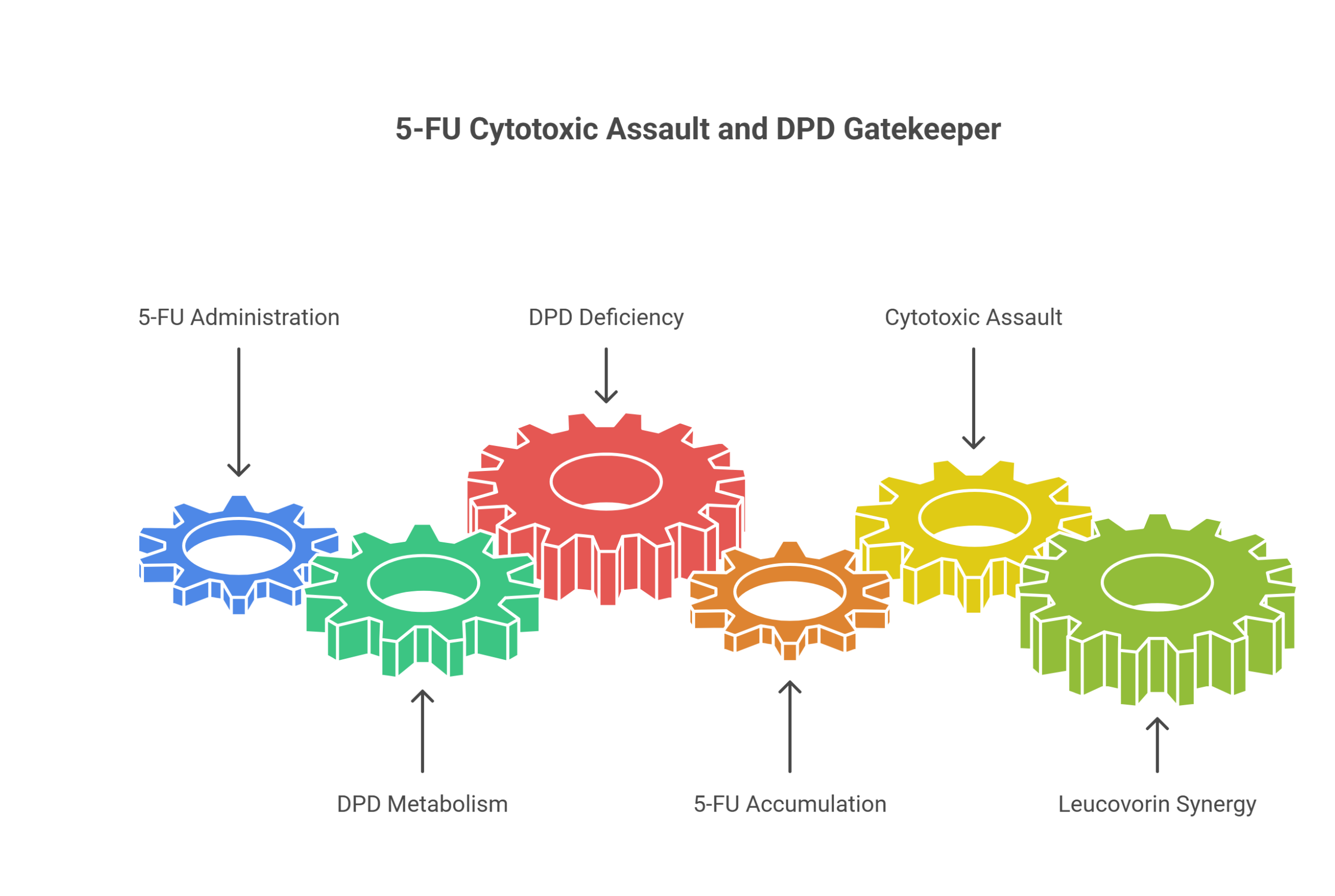
5-FU does not rely on a single mechanism. Its metabolites wage a three-pronged cytotoxic assault: FdUMP inhibits thymidylate synthase, FUTP incorporates into RNA and disrupts protein synthesis, and FdUTP lodges into DNA, causing strand breaks. Bolus administration tends to favor the RNA pathway; continuous infusion shifts the weight toward TS inhibition — which is precisely why leucovorin’s synergy matters most in infusion-based protocols.
But before any of this can happen, the drug must survive its own destruction. An enzyme called dihydropyrimidine dehydrogenase — DPD — metabolizes roughly 80% of administered 5-FU into inactive metabolites. In patients with DPD deficiency, this gatekeeper fails. Standard doses become catastrophically toxic — and adding leucovorin on top supercharges a drug the body already cannot clear.
The Protocols: Precision as Medicine
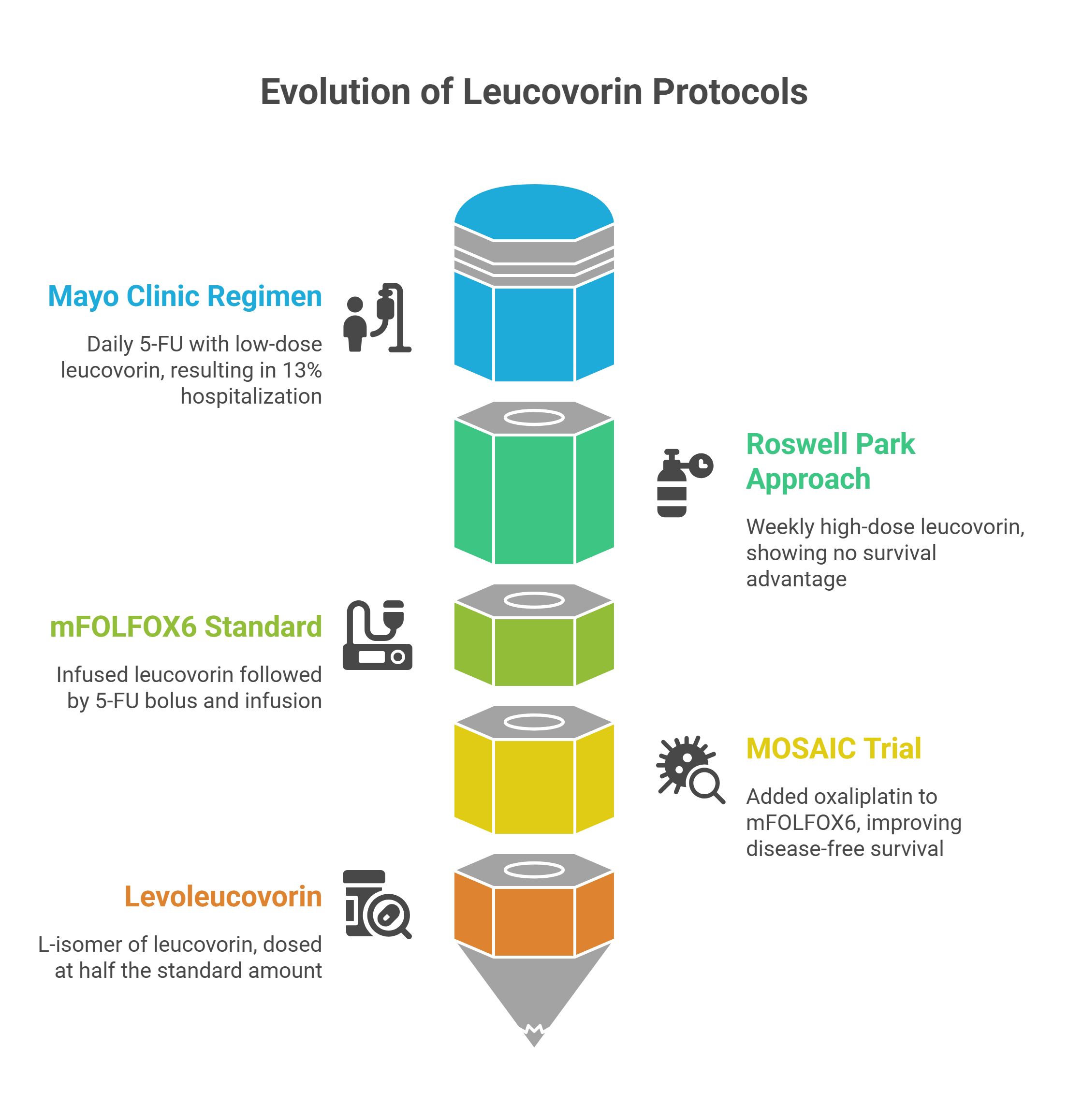
The evolution of leucovorin-enhanced regimens tells the story of oncology learning, sometimes painfully, how to calibrate this partnership. The Mayo Clinic regimen — leucovorin 20 mg/m² with 5-FU 425 mg/m² daily for five days, repeated every four weeks — carried a 13% hospitalization rate for chemotherapy-related toxicity. The Roswell Park approach swung the other direction, using 500 mg/m² of leucovorin weekly. Yet randomized trials showed no meaningful survival advantage for high-dose over low-dose leucovorin.
The modern standard — mFOLFOX6 — threads the needle. Leucovorin 400 mg/m² is infused over two hours, followed by a 5-FU bolus and a 46-hour continuous infusion. The leucovorin must be on board before the 5-FU arrives; reverse the sequence, and you miss the synergy window entirely. The MOSAIC trial demonstrated that adding oxaliplatin to this fluorouracil-leucovorin backbone significantly improved disease-free survival in stage III colon cancer.
A critical safety distinction sits inside those pharmacy orders. Standard leucovorin is a racemic mixture; only the L-isomer is biologically active. Levoleucovorin is dosed at exactly half — 200 mg/m² instead of 400. Confuse the two, and you have doubled the active folate in a patient’s system, a potentially lethal miscalculation.
When the Wheels Come Off
The same mechanism that makes leucovorin so effective against tumors makes it devastating to healthy tissue. The gut lining and bone marrow — tissues that divide relentlessly — absorb the enhanced damage. Toxicity-related hospitalizations occur in 20–30% of treated patients, with dose-limiting mucositis, neutropenia, and diarrhea.
Overexposure — from overdose, DPD deficiency, or drug interactions — can trigger severe myelosuppression, GI hemorrhage, septic shock, and multi-organ failure. And here is where a common, dangerous misconception lurks: leucovorin cannot rescue a patient from 5-FU toxicity. Giving it would only intensify the crisis.
The actual antidote is uridine triacetate (Vistogard), FDA-approved in 2015. It works by flooding cells with uridine, which competes with toxic FUTP for incorporation into RNA. In a 173-patient study, 94% of patients treated with uridine triacetate recovered fully within 30 days — compared to approximately 90% mortality in historical controls receiving only supportive care.
The Prodrug Twist
The quest to make 5-FU smarter led to capecitabine, an oral prodrug that doesn’t become active until it passes through a three-step enzymatic conversion. The final step relies on thymidine phosphorylase, an enzyme found at higher concentrations in tumor tissue — an elegant attempt at tumor-selective activation. But once capecitabine awakens as 5-FU inside the cell, it faces the same folate-limiting bottleneck. The ternary complex still needs its cofactor. The fundamental biochemistry remains unchanged.
The Volume Knob
Leucovorin is not a vitamin supplement. It is a precision instrument — the volume knob on one of oncology’s most powerful amplifiers. Turn it to the right setting, and a cancer drug with an 11% response rate becomes one that responds in nearly a quarter of patients. Turn it too high, confuse the isomer, or administer it in the wrong context, and you transform a treatment into a crisis. In the distance between those two outcomes lies everything that modern oncology has learned about the molecular choreography of killing cancer while keeping the patient alive.