Psychological Factors in Obsessive Relationships: Clinical Considerations for Healthcare Professionals
April 7, 2026
The Vitamin That Makes Chemotherapy Deadly
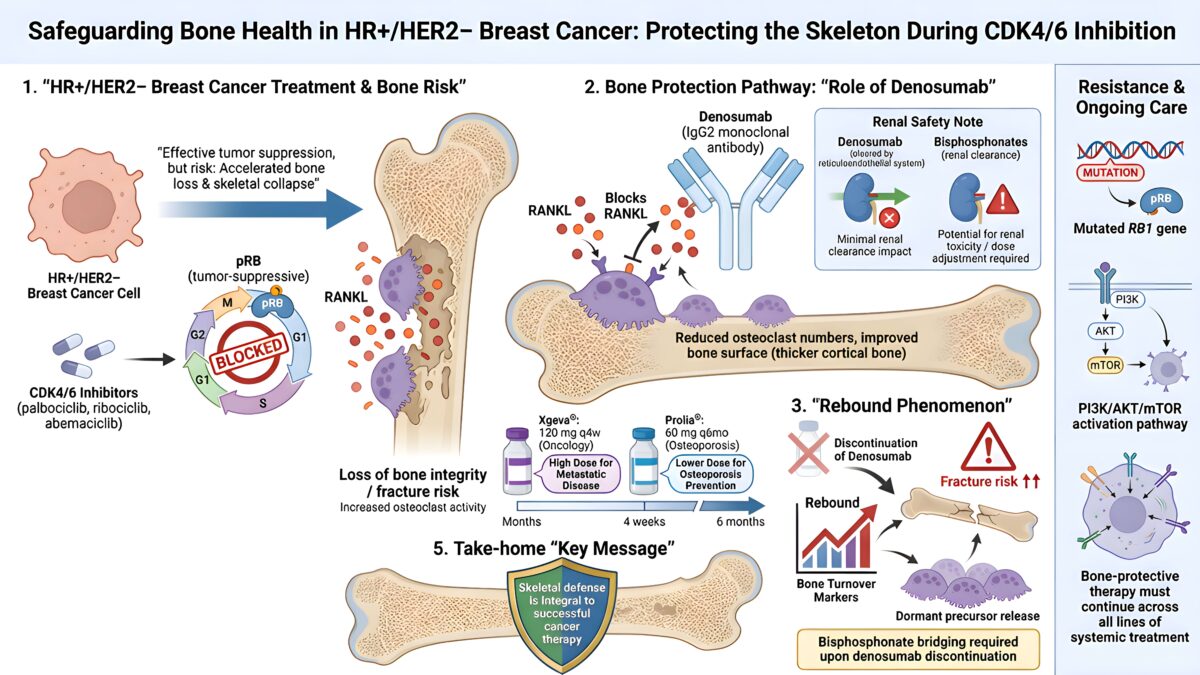
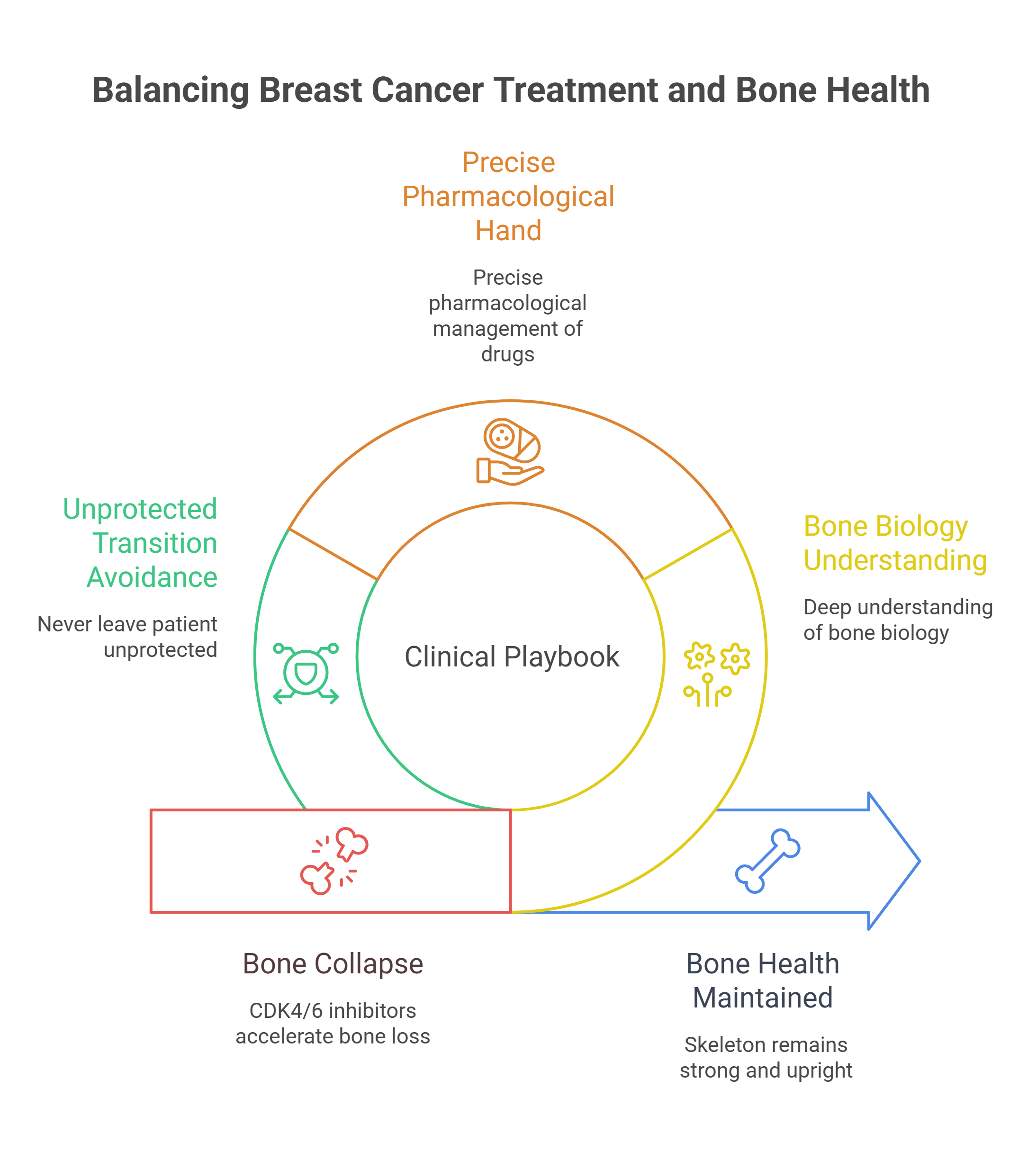
April 21, 2026Three FDA-approved CDK4/6 inhibitors — palbociclib, ribociclib, and abemaciclib — have reshaped the first-line treatment of HR+/HER2− breast cancer. They halt the cell cycle by blocking the G1-to-S phase transition, keeping the retinoblastoma protein in its tumor-suppressive state. But these agents almost always travel alongside aromatase inhibitors, and that pairing creates a paradox: the very regimen slowing the cancer is accelerating the collapse of the skeleton. Managing this tension requires a deep understanding of bone biology, a precise pharmacological hand, and the discipline never to leave a patient unprotected during a treatment transition.
The RANKL Axis: Where the Bone Battle Begins
Denosumab is a fully human IgG2 monoclonal antibody engineered to intercept a single molecular conversation. Its target — RANKL — is a soluble trimer belonging to the TNF superfamily. When RANKL binds its receptor RANK on the surface of hematopoietic precursors, it triggers the differentiation, activation, and prolonged survival of osteoclasts, the cells that dissolve bone. Denosumab competitively blocks this binding, preventing osteoclast maturation and promoting the apoptosis of existing osteoclasts. In effect, it mimics the body’s own decoy receptor, osteoprotegerin, but with far greater potency and reliability.
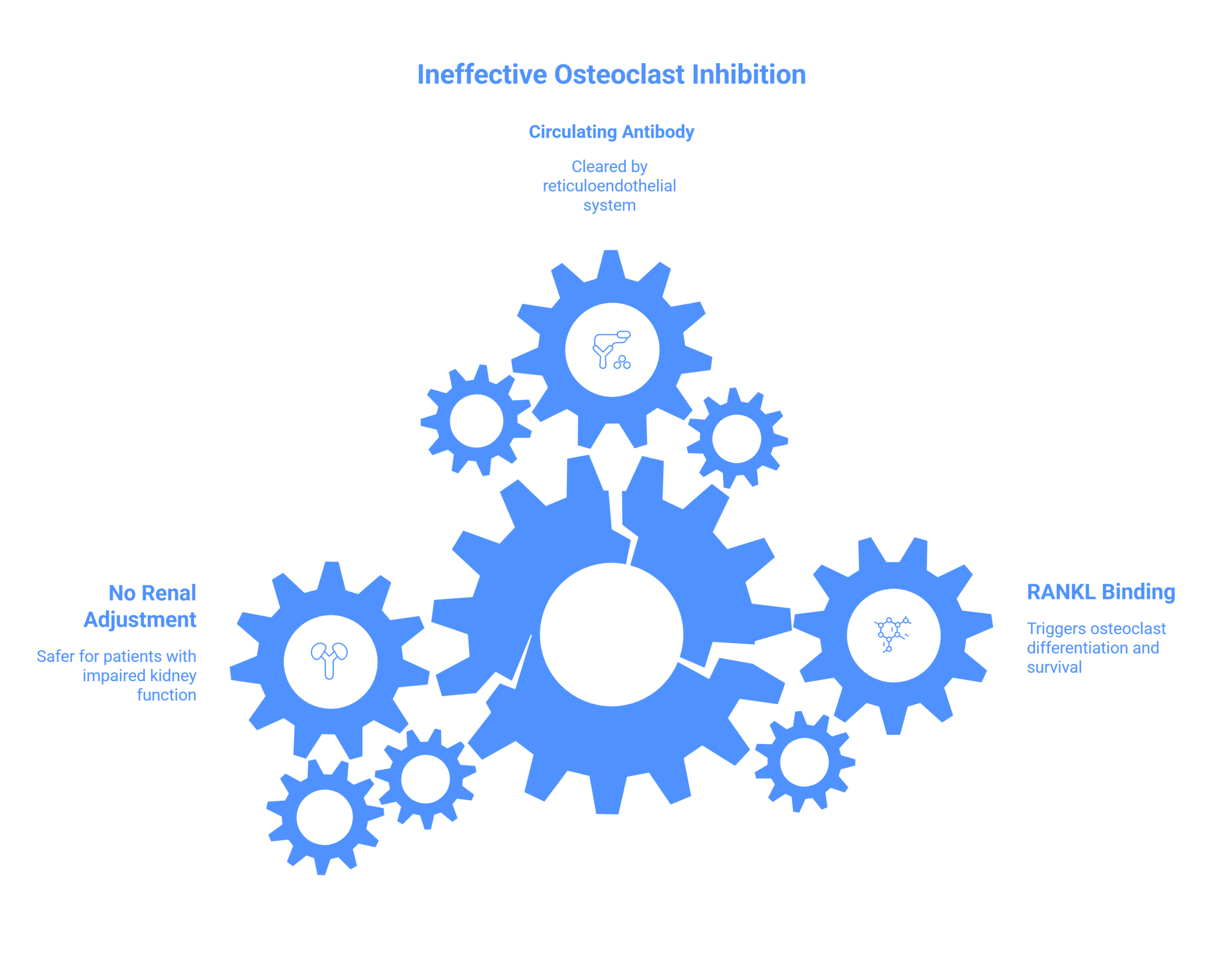
Unlike bisphosphonates, which embed themselves in the mineral matrix and persist for years, denosumab is a circulating antibody. It is cleared by the reticuloendothelial system, not the kidneys. This pharmacokinetic distinction carries enormous clinical weight: denosumab requires no dose adjustment in renal impairment, making it the safer choice for patients whose kidney function has deteriorated under the cumulative burden of cancer therapy.
Xgeva in Oncology: The Dosing Distinction That Matters
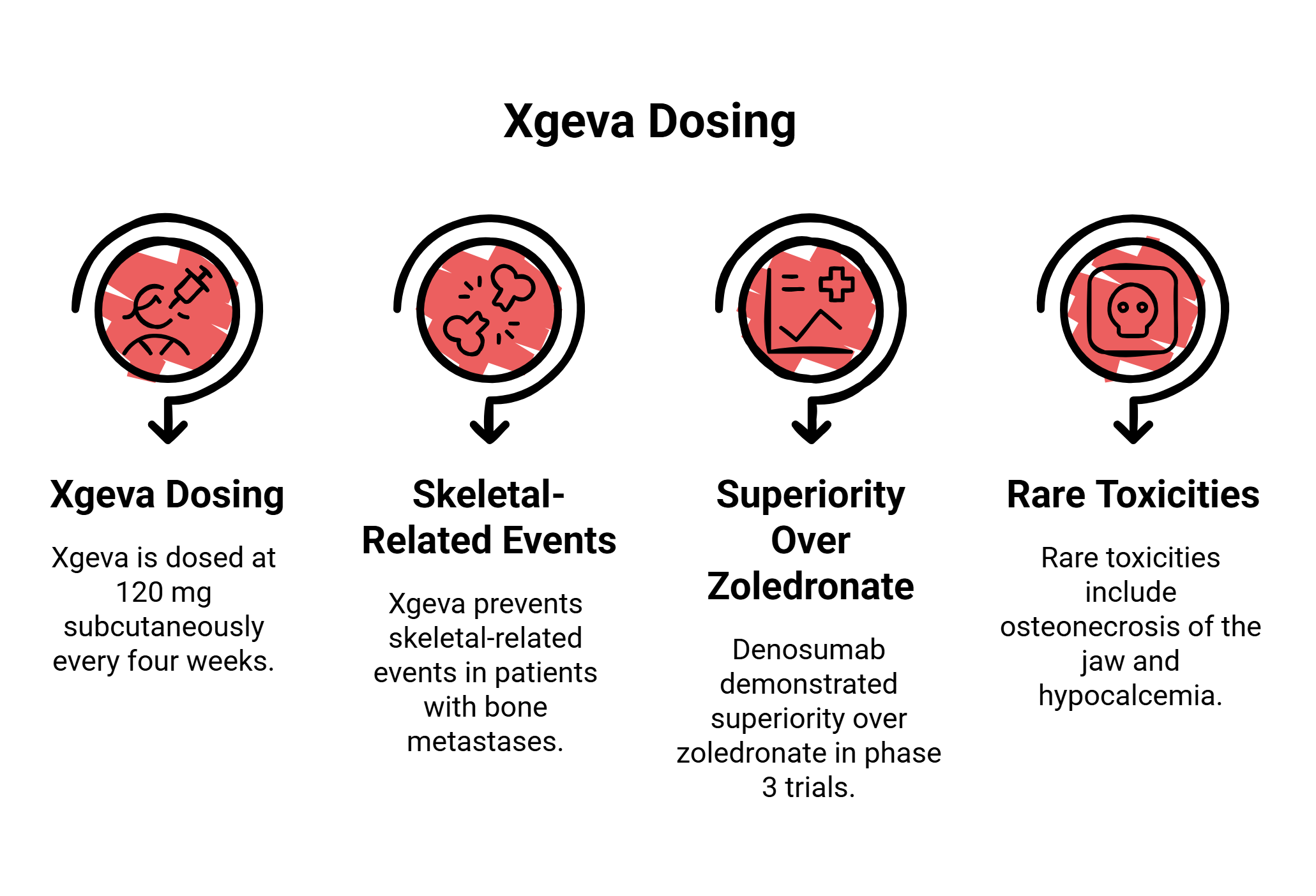
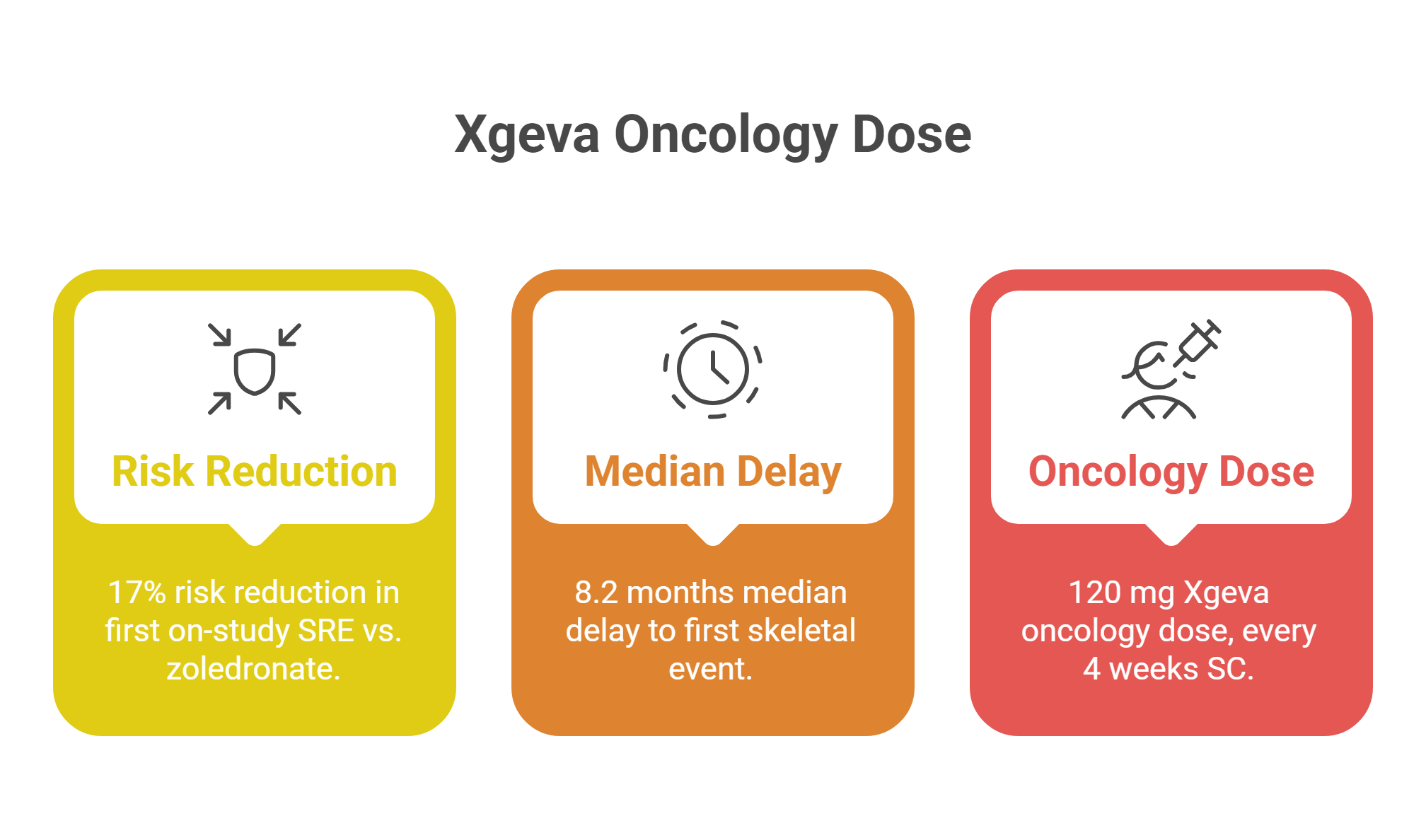
A critical distinction separates the two commercial faces of denosumab. Prolia — 60 mg every six months — is the osteoporosis formulation. Xgeva is dosed at 120 mg subcutaneously every four weeks for the prevention of skeletal-related events in patients with bone metastases from solid tumors and multiple myeloma. Conflating these schedules is a clinically dangerous error. In the phase 3 trials that secured FDA approval, denosumab demonstrated superiority over zoledronate, reducing the risk of first on-study SRE by 17 percent and the risk of first-and-subsequent events by 18 percent across multiple solid tumor types.
Rare but serious toxicities include osteonecrosis of the jaw, hypocalcemia, and atypical femoral fractures — risks that scale with duration and demand vigilant dental monitoring and calcium supplementation throughout treatment.
The Rebound Phenomenon: Why You Can Never Just Stop

Data from the landmark FREEDOM trial revealed a phenomenon that should unsettle every prescriber. When denosumab is discontinued, bone turnover markers begin climbing within three months of a missed dose. By six months, they exceed pre-treatment levels. By twelve months, all bone mineral density gains are erased. Vertebral fracture risk surges over the subsequent six to eighteen months.
The mechanism is disturbingly logical. Because denosumab does not induce osteoclast apoptosis in the way bisphosphonates do but instead arrests osteoclast development in a highly reversible manner, clearance of the drug unleashes a dormant pool of osteoclast precursors. RANKL expression surges, and the skeleton is subjected to a wave of resorption that can be more destructive than the original disease state. One systematic review of 24 cases documented an average of 4.7 vertebral fractures per patient only 5.2 months after the drug’s effect should have dissipated.
The clinical consensus is clear: if denosumab is stopped, patients must be bridged onto a bisphosphonate — typically a single dose of zoledronic acid six months after the last denosumab injection, or at least one year of oral alendronate. Serum CTX and BMD should be monitored, and re-dosing considered if markers remain elevated.
When the Cancer Outmaneuvers CDK4/6 Inhibitors
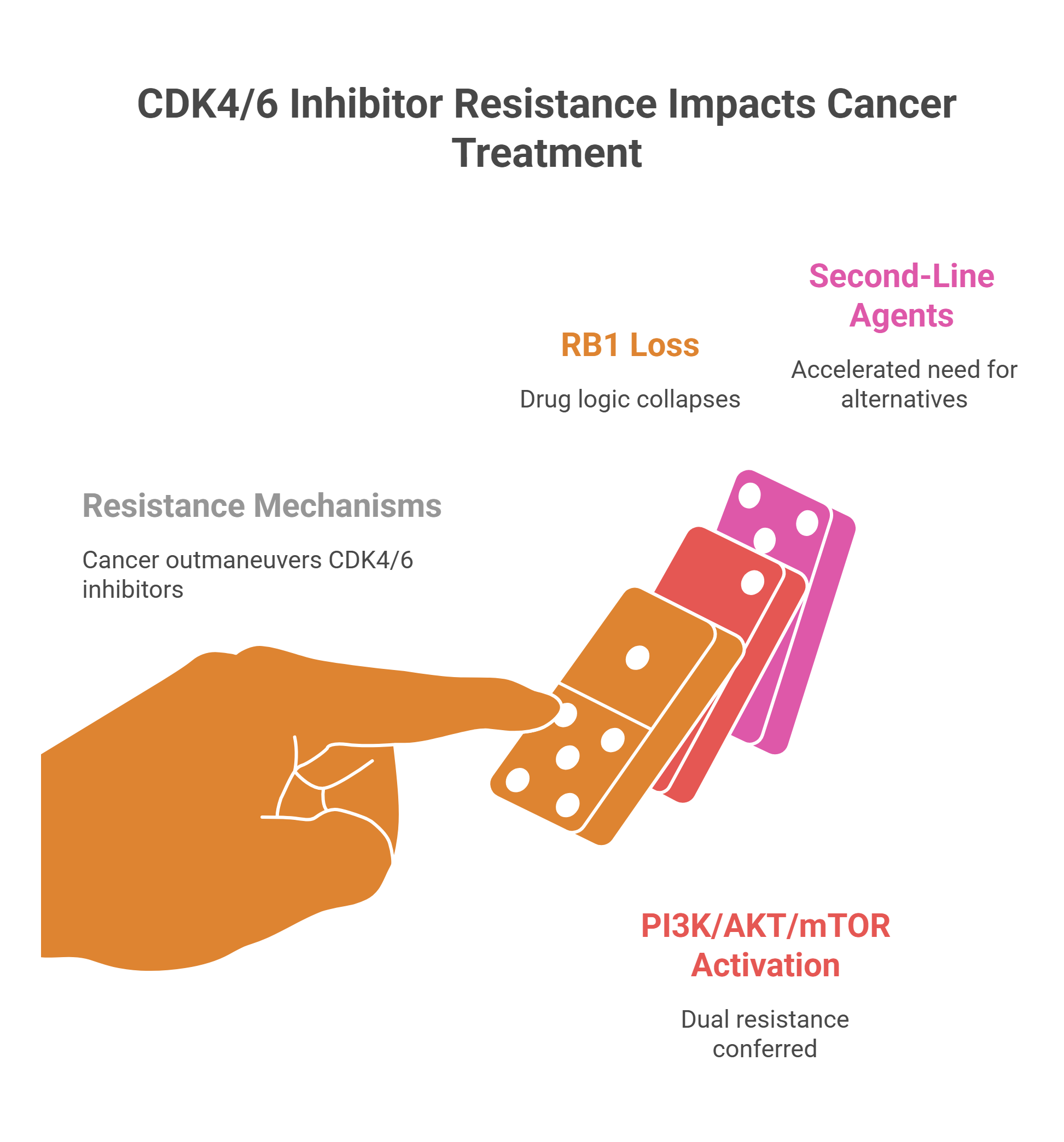
Resistance to CDK4/6 inhibitors is nearly inevitable, and understanding its architecture is essential for protecting both the tumor response and the skeleton. Loss of the RB1 gene is among the most consequential mechanisms: without retinoblastoma protein, the drug’s entire logic collapses. The cell cycle proceeds through activation of E2F transcription factors and the Cyclin E–CDK2 axis, rendering CDK4/6 blockade irrelevant.
Equally potent is the activation of the PI3K/AKT/mTOR signaling cascade, most commonly driven by PIK3CA mutations found in 30 to 40 percent of HR-positive breast cancers or by PTEN loss. These aberrations confer dual resistance to both CDK4/6 inhibitors and endocrine therapies, accelerating the need for second-line agents — elacestrant for ESR1 mutations, alpelisib for PIK3CA-driven disease, trastuzumab deruxtecan for HER2-low tumors, or sacituzumab govitecan targeting TROP-2. Throughout every line switch, bone-protective therapy must remain uninterrupted.
The Skeleton Is Not a Side Issue
Modern oncology has become extraordinarily good at keeping tumors in check. But every month a patient survives on aromatase inhibitors and CDK4/6 blockade is another month of cumulative skeletal assault. Denosumab — dosed precisely, monitored relentlessly, and never abruptly withdrawn — is the instrument that preserves the structural foundation on which all other therapies depend. The bone is not a bystander. It is the ground the patient stands on, literally and therapeutically. Defend it with the same urgency you bring to the cancer itself.