
Overview of CNS Neurotransmitters
November 16, 2021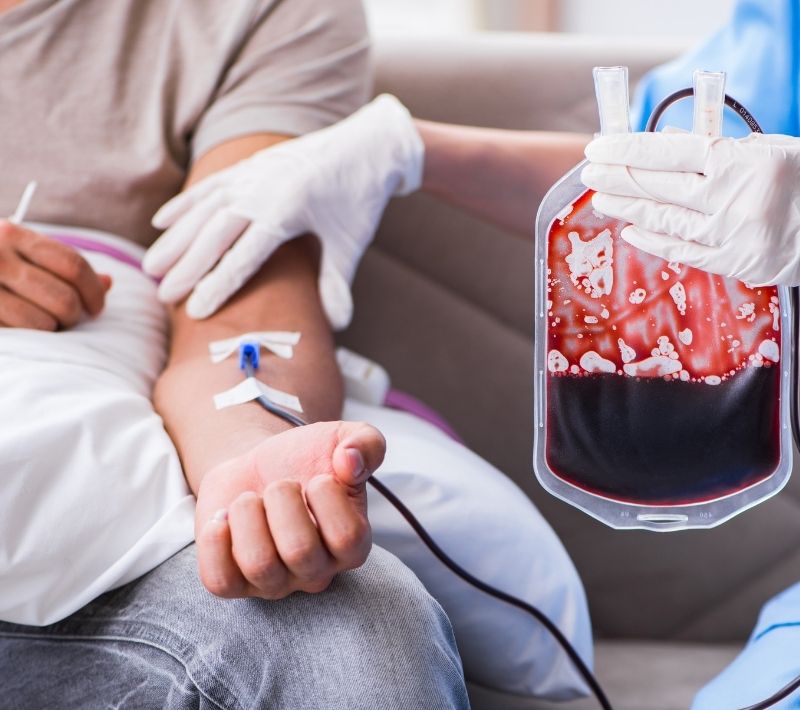
Blood transfusions are not optimal in the treatment of anemia
November 16, 2021Prevalence of ID (Iron Deficiency) & IDA (Iron Deficiency Anemia).
Iron deficiency anemia
Disease progression
Anaemia exacerbates underlying disease in IBD,3 and increases the risk of disease progression in CKD and CHF4,5
- greater risk of mortality, morbidity 6–8
- Increased fatigue and decreased QoL9
Hospitalizations
We can associate IDA with hospitalization of patients or prolongation of hospital stay1-3, 6-8
- Anemia increases the costs of managing CKD-ND patients by increasing patients’ risk of hospitalization and by hastening their progression to dialysis2.
- We see increased healthcare costs in CHF and cancer patients with anemia, primarily driven by higher rates of hospitalisation3,4.
Blood transfusion
Pre-operative IDA can be associated with increased blood transfusions
- Blood transfusions are frequently used to correct severe anemia, yet they are associated with increased mortality, morbidity, and length of hospitalisation4–6 and have significant costs13,14.
- Kovesdy CP et al. Kidney Int 2006;69:560–564;
- Caro JJ et al. Cancer 2001;91:2214–2221;
- Kulnigg S, Gasche C. Aliment Pharmacol Ther 2006;24:1507–1523;
- Silverberg D. Nephrol Dial Transplant 2003;18 Suppl 2:ii7–12;
- Wexler D et al. Nephrol Dial Transplant 2005;20 Suppl 7:vii11–vii15;
- Rossert J et al. Kidney International 2005;68:S76–S81;
- WHO. 1999. Available at: http://whqlibdoc.who.int/publications/1999/9241561955_eng.pdf;
- Morrison J et al. J Reprod Med 2008;53:323–330;
- Cohen BJ, Gibor Y. Obstet Gynecol Surv 1980;35:597–618;
- Vogelzang NJ et al. Semin Hematol 1997;34:4–12;
- Gasche C et al. Gut 2004;53:1190–1197;

ID/IDA in the life course of women

Most common causes of ID/IDA
Decreased iron intake
- Vegetarian or otherwise unbalanced diet4
- Eating disorder5
- Disease-related anorexia6
Decreased iron absorption
- Coeliac disease7
- Malabsorption2
- Chronic inflammatory or malignant diseases2
- Concomitant intake of drugs2
Blood loss
- Heavy or prolonged menstrual bleeding (HMB)1,2
- Delivery2,3
- Gastrointestinal bleeding2
- Surgery2
- Blood donation2
Increased iron demand
- Pregnancy and lactation2–4
- Infancy2,4
- Adolescence2,4
- Endurance sport8
- Marret et al. Eur J Obst Gyn Reprod Biol 2010;152:133–7
- Crichton et al. UNI-MED Verlag AG, 2008
- Milman. Ann Hematol 2006;85:559–65
- Zimmermann & Hurrell. Lancet 2007;370:511–20
- Barton et al. BMC Blood Disorders 2010;10:9
- Aapro et al. Ann Oncol 2012;23:1954–62
- Goodnough. Transfusion 2012;52:1584–92
- Stein et al. Nat Rev Gastroenterol Hepatol 2010;7:599–610
Prevalence of anemia – pregnancy

ID/IDA World Health Organization statements
All women of reproductive age are at risk of iron deficiency. It is estimated that 30% of women globally are anemic, with at least half of these cases arising from iron deficiency.
Among pregnant women, IDA has been associated with increased risks of low birth weight, prematurity, and maternal morbidity.
Iron deficiency and anemia reduce the work capacity of individuals and entire populations, bringing serious economic consequences and obstacles to national development.
- WHO. Preconception Care to Reduce Maternal & Childhood Mortality & Morbidity. 2012. http://apps.who.int/iris/bitstream/10665/78067/1/9789241505000_eng.pdf
- WHO. Guideline: Daily iron and folic acid supplementation in pregnant women. 2012. http://apps.who.int/iris/bitstream/10665/77770/1/9789241501996_eng.pdf
- WHO. Micronutrient deficiencies. 2013. http://www.who.int/nutrition/topics/ida/en/
Anemia in women
Heavy menstrual bleeding is the leading cause of iron deficiency/iron deficiency anemia in women.

Women have an increased iron need
- The average iron content of the body is 2500–4000 mg, with women having a lower body iron content than men1,2
- Most body iron (~1800 mg) is contained as hemoglobin in circulating red blood cells. Virtually all of the iron released after the breakdown of red blood cells is reused, and only 1–2 mg of iron is lost per day through shedding of cells in the gut or skin or menstrual blood loss1
- Non-pregnant women require 1.5–2 mg iron/day from their diet to compensate for this loss of body iron compared with 1.0 mg iron/day for men1
- Women are estimated to have a median dietary iron intake of 12 mg/day, which is lower than their RDA of 18 mg/day. The evidence of insufficient iron intake suggests that the additional iron demand from menstrual blood loss is not being met3

- Crichton R et al. Iron therapy with special emphasis on intravenous administration. Uni-Med Verlag AG, London, Boston 2008.
- Hentze MW et al. Cell 2004:117:285‒297.
- Institute of Medicine, Food and Nutrition Board. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. National Academies Press, 2001:1–28. Available at: http://www.iom.edu/Reports/2001/Dietary-Reference-Intakes-for-Vitamin-A-Vitamin-K-Arsenic-Boron-Chromium-Copper-Iodine-Iron-Manganese-Molybdenum-Nickel-Silicon-Vanadium-and-Zinc.aspx
Clinical definition of HMB
For clinical purposes, HMB should be defined as excessive menstrual blood loss which interferes with the woman’s physical, emotional, social and material quality of life, and which can occur alone or in combination with other symptoms. HMB should be recognized as having a major impact on a woman’s quality of life.
Any interventions should aim to improve quality-of-life measures.
NICE guideline. Heavy menstrual bleeding. 2007. http://www.nice.org.uk/nicemedia/live/11002/30404/30404.pdf
Consequences of HMB associated with ID1–4

- There is a clear association between excessive menstrual blood loss and ID in women and adolescent girls1,2
- As a result, several consequences of HMB are also causally related to ID1,3-6
- Associations between HMB-associated factors (in particular, fatigue and impaired cognitive function) and ID are exemplified by improvements in these symptoms following iron supplementation3,5
- Wang W et al. Haemophilia 2013;19:225-30.
- Al-Quaiz JM. Saudi Med J 2001;22:490-6.
- Verdon F et al. BMJ 2003;326:1124.
- Brownlie T 4th et al. Am J Clin Nutr 2004;79:437-43.
- Bruner A et al. Lancet 1996;348:992–996.
- Liu Z et al. Value Health 2007;10:183–94.
General consequences of ID/IDA
It is well established that ID/IDA exerts various negative effects1,2. Specifically, ID/IDA has been shown to adversely influence physical and mental performance (exercise, physical activity, fatigue, mental and cognitive ability and performance)1,3,4 physiological processes (enzymatic activity such as the respiratory chain; thermoregulation)1 and immune (potential susceptibility to infections) and neurological function1. ID/IDA also affects the ability to tolerate heavy blood loss5

- Breymann C. Fetal Matern Med Review 2002;13:1–29.
- Milman N. Ann Hematol 2008;87:949–959.
- Rowland TW et al. Am J Dis Childhood 1988;142:165–169.
- Bruner A et al. Lancet 1996;348:992–996.
- Hercberg S et al. Clin Drug Invest 2000;19(Suppl 1):1–7.
Quality of life improvement in women with HMB after 5 years

How to manage ID/IDA

- This schematic treatment algorithm illustrates recommended hemoglobin and serum ferritin thresholds dictating a diagnosis of ID/IDA and subsequent management in pre-menopausal (non-pregnant) women.1
- Oral iron supplements at doses of 100 to 200 mg iron/day (IDA) or 60 to 100 mg iron/day (ID) are recommended as first-line treatment for women, with dosage adjusted according to ongoing diagnostic assessment.1
- IV iron is advised as an alternative in cases of non-response or severe IDA; to get accurate measurements of treatment effect, serum ferritin levels should be assessed 12–16 weeks after initiation of IV treatment.1
- Recent guidance from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) also recommends measures to minimize the risk of allergic reactions with IV iron use.2
- Breymann C et al. Geburtsh Frauenheilk 2013;73:256-261.
- European Medicines Agency. New recommendations to manage risk of allergic reactions with intravenous iron-containing medicines. 2013.
How to diagnose ID/IDA1
- Normal hemoglobin levels vary with age, sex and physiological status, e.g. during pregnancy. Anemia occurs when hemoglobin levels fall below. For example, the WHO recommended threshold1
- Since conditions other than ID, e.g. vitamin B12 or folate deficiency, can also cause anemia, definitive diagnosis of IDA includes laboratory evidence of ID,2 such as levels of the following parameters:
- A low serum ferritin value shows absolute ID, although levels may not solely reflect iron stores in all cases.3 Serum ferritin is also an acute-phase reactant that is elevated, independently of body iron, in response to inflammatory processes4
- TSAT reflects the availability of iron in the plasma,4 and is the percentage of occupied iron-binding sites on transferrin which is presented as the ratio of serum iron and total iron-binding capacity (TIBC)
- Serum (or soluble) transferrin receptor (sTfR) is the serum concentration of transferrin receptor shed from reticulocytes; it shows iron-deficient erythropoiesis and tissue iron deficiency, and is unaffected by inflammation.5 In ID, the numbers of transferrin receptors increase significantly,4 but sTfR is the only diagnostic marker of ID that appears to remain at normal levels during pregnancy6
- de Benoist B, McLean E, Egli I et al. Worldwide prevalence of anaemia 1993-2005: WHO global database on anaemia. 2008. Available at: http://whqlibdoc.who.int/publications/2008/9789241596657_eng.pdf.
- Breymann C & Huch R. UNI-MED 2008:13–96
- Guyatt GH, Oxman AD, Ali M, et al. Laboratory diagnosis of iron-deficiency anemia: an overview. Journal of General Internal Medicine 1992;7(2):145–53.
- Crichton R, Danielson BG, Geisser P et al. Iron therapy with special emphasis on intravenous administration. Uni-Med Verlag AG, London, Boston 2008.
- Kohgo Y et al. Int J Hematol 2002;76:213–18
- Bothwell TH. Am J Clin Nutr 2000;72:257S–264S.

Medical need and treatment of Iron Deficiency Anemia

- 1. Breymann & Huch. UNI-MED 2008:13–96
2. WHO. Worldwide prevalence of anaemia, 1993–2005. 2008. - McMurray et al. Eur J Heart Failure 2012;14:803–869
4. Kohgo et al. Int J Hematol 2002;76:213–218; 5. Guyatt et al. J Gen Intern Med 1992;7:145–153
Anemia has a profound effect on the quality of life. Therefore, normalisation of the Hb level and iron status is a goal in the overall management of underlying diseases (Van Assche 2013, McMurray 2012).
Notably, a retrospective analysis of patients with inflammatory bowel disease who were treated for anemia and achieved normal Hb levels showed a 50% recurrence after 10 months (Kulnigg 2009). Iron deficiency, defined as ferritin <30 μg/L, recurred within a median time of 19 months, and iron deficiency recurred faster in patients with a post-treatment ferritin level <100 μg/L (median 4 months) than in patients with ferritin levels between 100-400 μg/L (median 11 months) and ferritin >400 μg/L (median 49 months; p<0.001).
Sufficient iron repletion (ferritin 100-400 μg/L) can prevent or delay anemia recurrence (Evstatiev 2013, Kulnigg 2009).
Since over 90% of ingested iron remains unabsorbed, oral iron preparations frequently lead to the occurrence of GI adverse effects, including nausea, flatulence, diarrhea and gastric erosion (Van Assche 2013).
Animal and human studies show that the generation of reactive oxygen species (Fenton reaction) by non-absorbed iron might lead to the exacerbation of IBD.
Several studies have shown that I.V. iron is at least as effective as oral iron, delivers faster response rates, and is safer in all but a very few patients who may experience side-effects.
Intravenous iron therapy is advisable in the following cases: for iron-deficient patients who are intolerant or unresponsive to oral iron supplementation (i.e., those showing an insufficient increase in serum iron parameters within the first 2 weeks of treatment); for patients with severe anemia (Hb <10 g/dL); for patients with pronounced disease activity; and for patients who are being treated with erythropoiesis-stimulating agents. Patients with mild anemia (Hb >10 g/dL) can be adequately treated with 100 mg/day iron sulphate.
Limitations of Oral Iron Therapies1,2
Limited effectiveness – Impaired intestinal iron absorption
- Poor absorption (~90% not absorbed)
- Absorption and efficacy do not increase proportionally with higher doses. 2
- May not compensate for ongoing chronic blood loss1
- Long therapy, mostly, needs to be continued for 5-6 months or at for least 3 months after replenishment of iron stores
- Concomitant food or medication (e.g. phosphate binders, H2 blockers, proton pump inhibitors)
Compliance
- Frequent intolerance because of GI side effects
- Pill burden: usually 2 or 3 tablets per day
- Low dose (100mg/day) may help to mimic the GI side effects and improve tolerance
May be inadequate during ESA therapy
·Accelerated erythropoiesis can increase demand for iron beyond the amount supplied orally
Oxidative stress
High oral iron doses can saturate the iron transport system if it rapidly released the iron, resulting in oxidative stress
- Macdougall IC. Curr Med Res Opin 2010;26:473–83
- Crichton RR et al. Iron Therapy With a Special Emphasis on Intravenous Administration (4th edition). UNI-MED Verlag AG, Bremen, Germany, 2008.
- Kulnigg S, Gasche C. Aliment Pharmacol Ther 2006;24:1507–23
- Neil D, Clin and Exper Gastro, 2013,6, 61-70
Oral iron therapy for inflammatory bowel disease
Oral iron may not compensate for ongoing blood loss in IBD.1 Studies in animal models of inflammatory bowel disease (IBD) consistently show an increase in oxidative stress, disease activity, intestinal inflammation and colorectal cancer development through oral iron supplementation.2 This is not surprising, as approximately 90% of ingested iron is not absorbed, passes the sites of intestinal inflammation and induces local oxidative stress at sites of active inflammation (through the Fenton reaction).
Further studies show that nutritional iron may be one of the exogenous factors responsible for the onset of colitis.3,4 In patients with IBD, oral iron induces oxidative stress,5 increases local disease activity, particularly in patients with ulcerative colitis,6 and in patients with Crohn’s disease, the absorption of oral iron is inhibited, possibly through a hepcidin-mediated mechanism.7
If oral iron is used, there is limited data available to differentiate between products.
A study comparing iron polymaltose complex8 and ferrous sulphate showed that ferrous sulphate increased markers for lipid peroxidation more than iron polymaltose complex.
- Schreiber S et al. N Engl J Med 1996;334:619–23.
- Kulnigg S, Gasche C. Aliment Pharmacol Ther 2006;24:1507–23.
- Seril DN et al. J Clin Gastroenterol 2006;40:391–7.
- Lee KM, Sartor RB. Gastroenterology 2007;132:A701.
- Erichsen K et al. Scand J Gastroenterol 2003;38:543–8.
- De Silva AD et al. Aliment Pharmacol Ther 2005;22:1097–105.
- Semrin G et al. Inflamm Bowel Dis 2006;12:1101–6.
- Erichsen K et al. Aliment Pharmacol Ther 2005;22:831-33






1 Comment
[…] may advise pregnant women to stay active, avoid prolonged sitting or standing, and wear compression stockings to lower […]