Don’t Shake the Vial
May 1, 2026Imagine two surgeons who trained at the same school, mastered the same procedure, and operate on the same organ — yet whose techniques, tools, and outcomes diverge in ways that can mean months of extra life for a patient. That is precisely the situation oncologists face every time they sit down with a newly diagnosed case of RAS wild-type metastatic colorectal cancer and must choose between cetuximab (Erbitux) and panitumumab (Vectibix). Both drugs block the same receptor. Both are monoclonal antibodies. And yet, beneath that surface similarity lies a world of molecular, immunological, and clinical difference that increasingly matters at the bedside.
The Molecular Identity Crisis
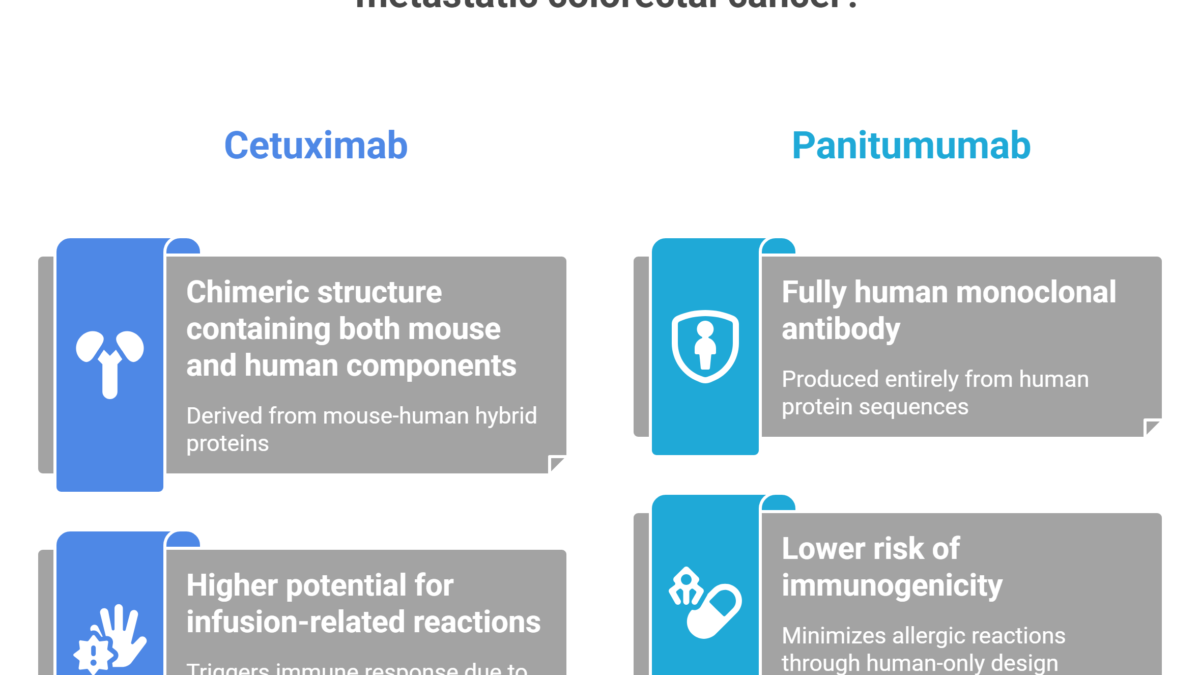
The story begins not in the clinic but in the molecular architecture of the drugs themselves. Cetuximab is a chimeric IgG1 monoclonal antibody — part mouse, part human. Its variable region, the targeting “head” of the molecule, is derived from murine cell lines. Panitumumab, by contrast, is a fully human IgG2 monoclonal antibody, produced through recombinant DNA technology so that not a single murine amino acid remains. As established in the landmark panitumumab versus cetuximab ASPECCT trial, this distinction is far more than a footnote in a pharmacology textbook.
Because cetuximab carries that murine component, the human immune system can recognize it as foreign. The result: a meaningfully higher rate of infusion-related reactions, ranging from chills and fever to full anaphylaxis. Panitumumab, presenting as entirely self to the immune system, sidesteps this problem almost entirely — eliminating the need for routine antihistamine premedication and significantly easing the infusion experience for patients and staff alike.
The IgG Subclass Divide
The IgG1 backbone of cetuximab confers a biological superpower that panitumumab’s IgG2 structure cannot replicate: Antibody-Dependent Cellular Cytotoxicity (ADCC). Cetuximab binds with high affinity to FcγRI and FcγRIIIa receptors on natural killer cells, effectively recruiting the immune system to destroy tumor cells. Panitumumab does not bind these receptors and has no ADCC activity — even though both drugs bind EGFR itself with comparable affinity. This has been confirmed in head-to-head biochemical studies indexed on PubMed (PMID 21178280) and reviewed comprehensively in Frontiers in Oncology.
The ASPECCT Verdict — And Its Nuances
For years, the clinical consensus rested on a single pivotal result: the two drugs are interchangeable. That conclusion came from the Phase III ASPECCT trial, published in Lancet Oncology, which enrolled chemotherapy-refractory patients with wild-type KRAS exon 2 metastatic colorectal cancer. The numbers were unambiguous — median overall survival of 10.4 months with panitumumab versus 10.0 months with cetuximab, producing a hazard ratio of 0.97 that crossed the non-inferiority threshold comfortably.
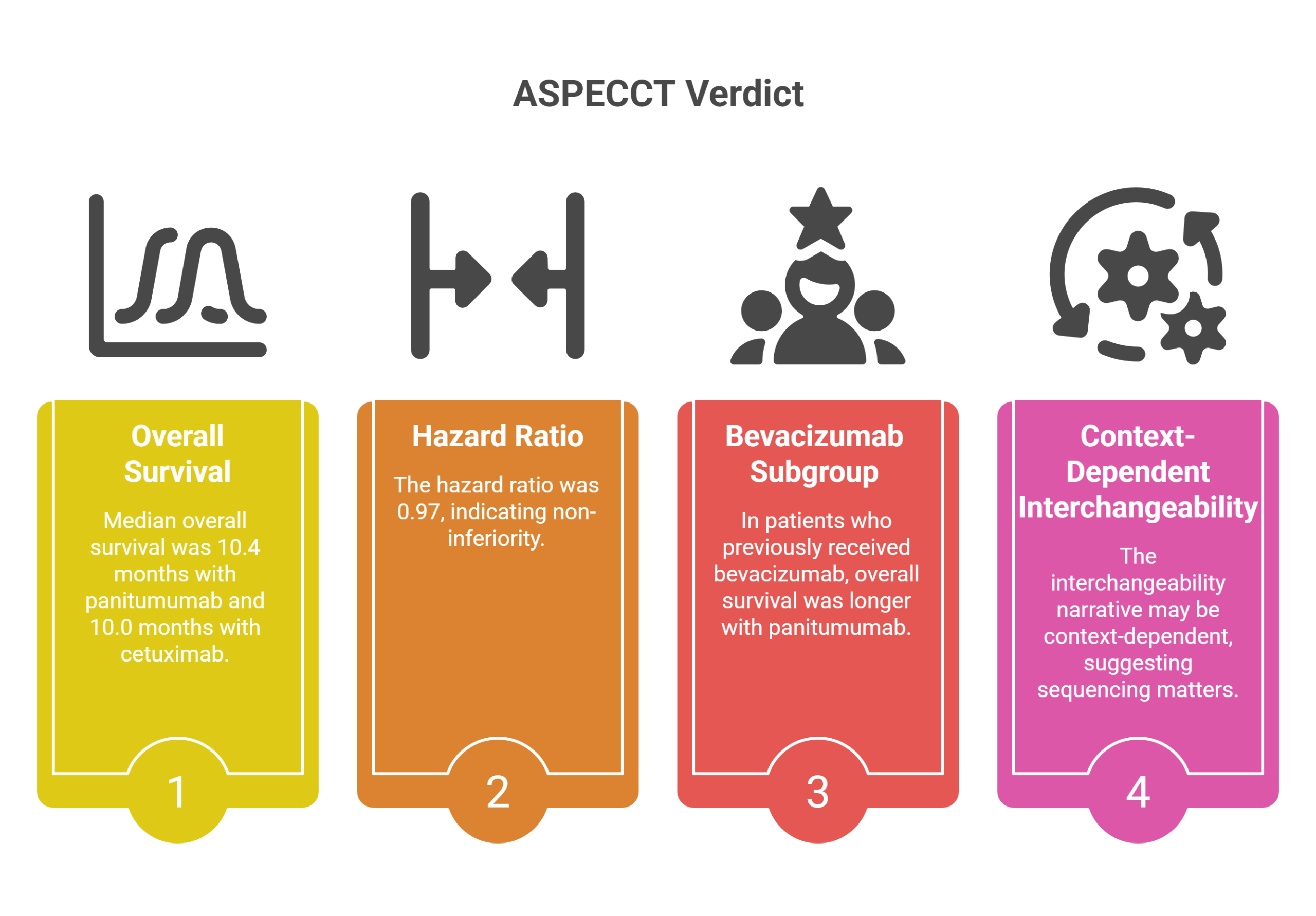
But the ASPECCT data held one buried finding that pointed toward something more complex. In patients who had previously received bevacizumab, overall survival was meaningfully longer in the panitumumab arm. This subgroup signal, now supported by additional Japanese real-world data, suggests the interchangeability narrative may be context-dependent — and that sequencing matters enormously.
The Tick Bite Nobody Expected
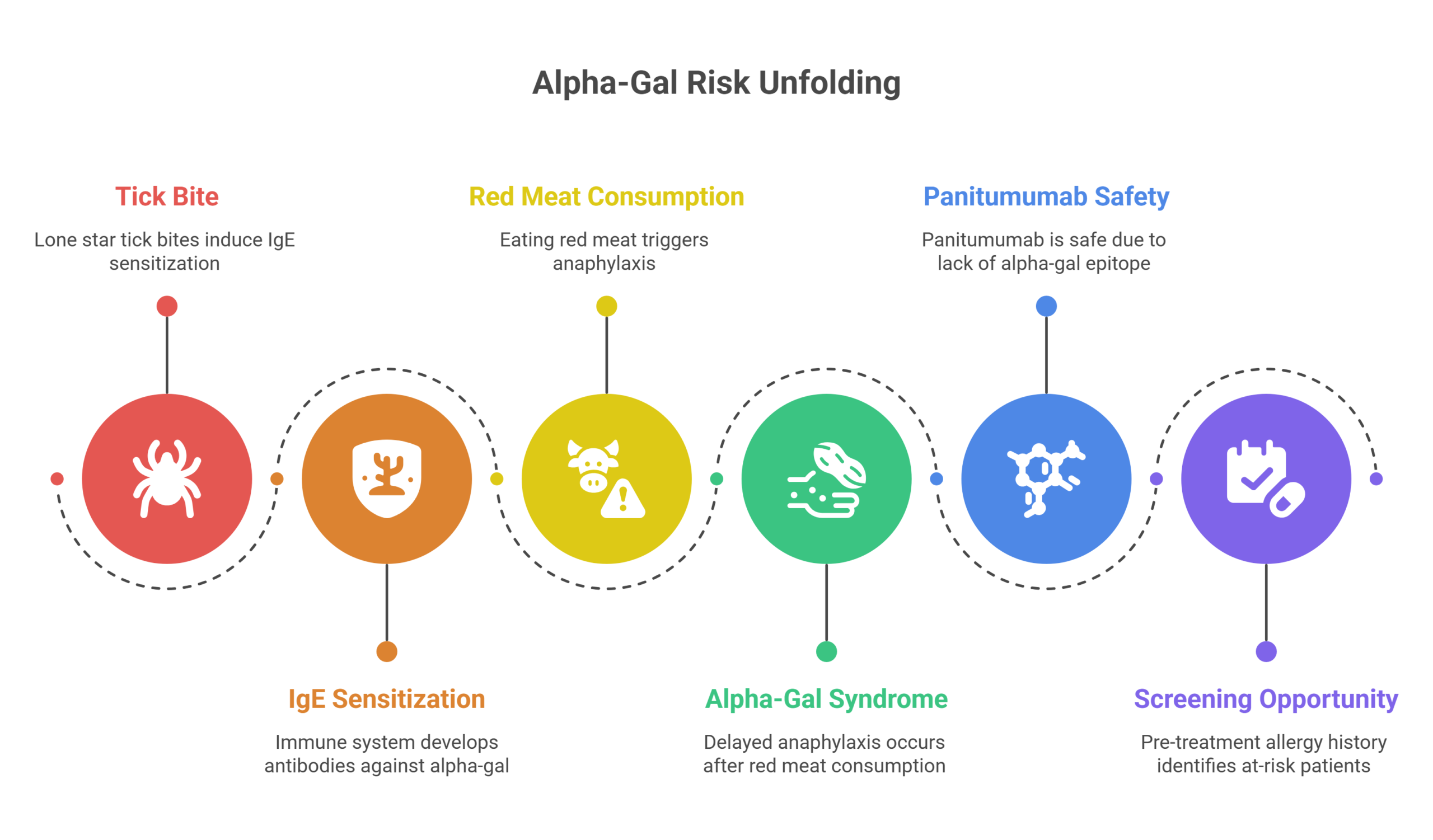
Perhaps the most striking finding in this entire clinical story has nothing to do with tumor genetics. It begins with a tick bite. Research published in the New England Journal of Medicine established that most severe hypersensitivity reactions to cetuximab are caused not by the drug’s murine component in the classical sense, but by preformed IgE antibodies to galactose-α-1,3-galactose — a sugar epitope, known as alpha-gal, that sits on the Fab portion of cetuximab’s heavy chain. Critically, these IgE antibodies are present in a patient’s blood before they ever receive a single infusion.
HOW THE ALPHA-GAL RISK UNFOLDS
The Source
In the United States, IgE sensitization to alpha-gal is induced by bites of Amblyomma americanum — the lone star tick — making geographic prevalence a real clinical variable.
The Double Danger
The same IgE antibodies also trigger delayed anaphylaxis 3–6 hours after eating red meat such as beef and pork — a phenomenon now recognized as alpha-gal syndrome.
Why Panitumumab Is Safe
Because panitumumab is fully human and produced without murine cell-line glycosylation, it carries no alpha-gal epitope and poses none of this risk — confirmed by Mayo Clinic Labs.
The Screening Opportunity
A simple pre-treatment allergy history — tick exposure, atopy, red meat reactions — can identify at-risk patients before any drug is administered.
The Silent Toxicity: Magnesium Wasting
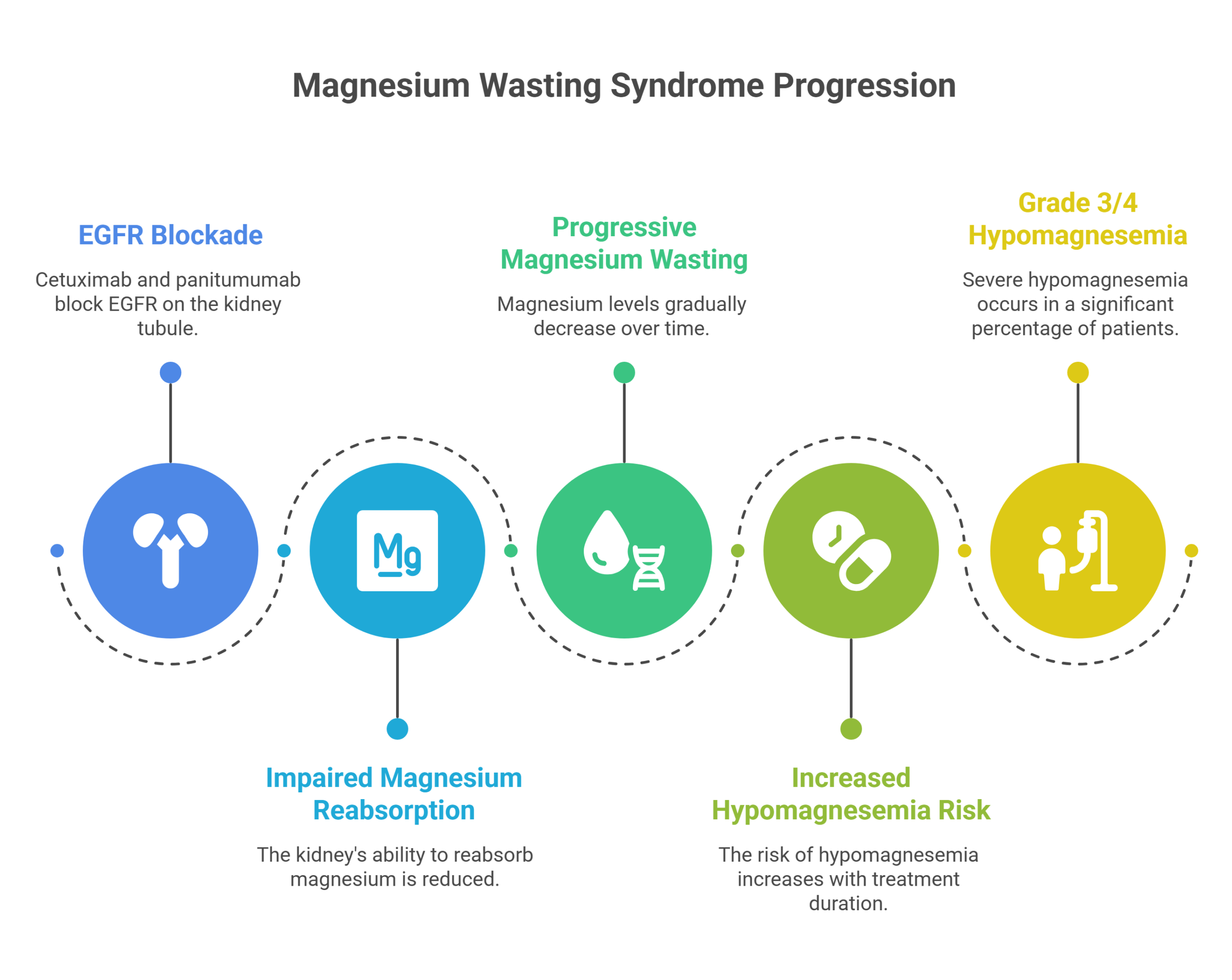
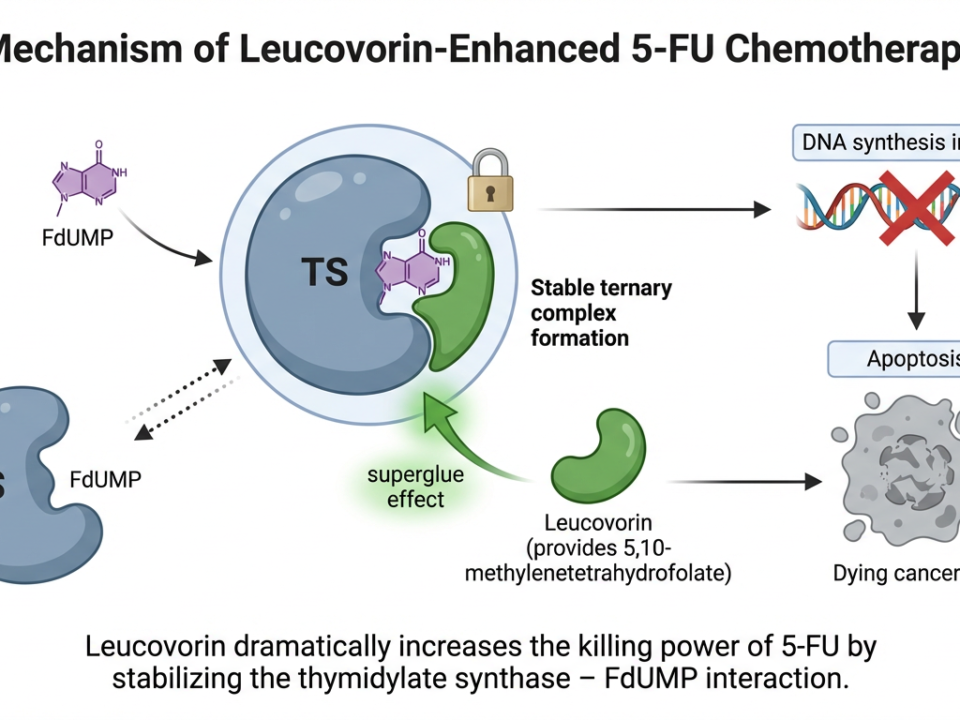
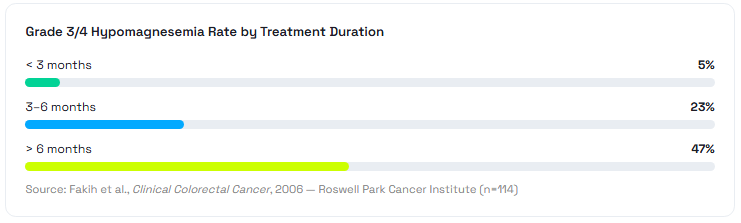
While the rash is the side effect patients see, hypomagnesemia is the one that can kill them quietly. Both cetuximab and panitumumab block EGFR on the distal convoluted tubule of the kidney, impairing the organ’s ability to reabsorb magnesium. The result is a progressive, cumulative magnesium wasting syndrome that worsens with every passing month of treatment. Data from Fakih et al. in Clinical Colorectal Cancer put the numbers in stark relief: grade 3 or 4 hypomagnesemia occurred in 5% of patients treated for fewer than three months — but climbed to 47% in those treated for more than six months.
Oral magnesium supplements fail to compensate — they cause diarrhea and cannot match the renal losses. The clinical answer is intravenous magnesium sulfate, sometimes required at doses of 6–10 grams per infusion, delivered multiple times weekly. Recovery typically occurs approximately four weeks after the anti-EGFR drug is discontinued, a fact that underpins the “stop-and-go” strategy used by experienced oncologists to balance tumor control with quality of life.
The BRAF Exception: When Cetuximab Takes Center Stage
For most RAS wild-type patients, the cetuximab-versus-panitumumab debate is at least a genuine contest. But for the roughly 8–12% of metastatic colorectal cancer patients who carry the BRAF V600E mutation, the playbook shifts dramatically — and here, cetuximab has become the entrenched partner of choice.
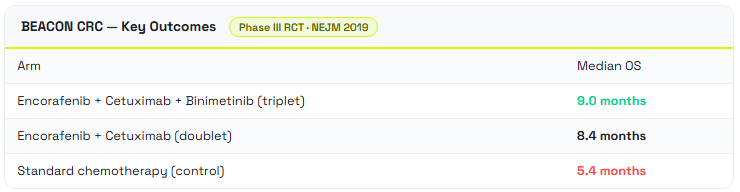
The reason is biological. When a BRAF inhibitor is administered alone, the tumor cell detects the drop in downstream signaling and compensates by dramatically upregulating EGFR — a feedback loop that effectively bypasses the block. Closing both the BRAF and EGFR doors simultaneously is the only strategy that works. The BEACON CRC trial in the New England Journal of Medicine proved the concept definitively: the combination of encorafenib, cetuximab, and binimetinib delivered a median overall survival of 9.0 months versus 5.4 months with standard therapy — a hazard ratio of 0.52 that was statistically unambiguous.
A 2025 systematic review and network meta-analysis published in The BMJ — covering 60 studies and 4,633 patients with BRAF-mutated advanced colorectal cancer — reinforced this direction, finding that combining doublet chemotherapy with anti-EGFR and BRAF-targeted agents offered the best overall survival benefit in the first-line setting.
The Decision Tree Is Not a Coin Flip
The era of treating cetuximab and panitumumab as therapeutic twins is giving way to a more nuanced, evidence-driven framework. Molecular subclass dictates immune risk. Tumor sidedness predicts anti-EGFR benefit. Prior bevacizumab exposure may tip the balance toward panitumumab. A tick bite in the patient’s history could turn a first infusion of cetuximab into a medical emergency. And for the unfortunate minority carrying BRAF V600E, cetuximab’s role is now defined by a landmark trial whose data is unambiguous.
What unites all of this is a principle that the best oncologists have always understood: knowing which drug to prescribe is only the beginning. Knowing why — down to the IgG subclass, the distal convoluted tubule, and the patient’s history with the lone star tick — is what separates standard care from optimal care. In metastatic colorectal cancer, that difference can be measured in months of life.