Revolutionary Swiss-Designed Technology Restores Mobility for Parkinson’s Patients
November 8, 2023
Comparing VTE Prophylaxis Medications for Deep Vein Thrombosis & Pulmonary Embolism
November 15, 2023Treatment of Gout and ULT Options GOALS
By the end of this article, you should be able to:
- Examine the pathophysiology and causes of gout, along with the signs and symptoms of acute attacks.
- Compare and contrast recommendations from various organizations’ guidelines for the pharmacological treatment of gout.
- Determine suitable treatment options for acute gouty arthritis and ongoing urate-lowering therapy.
- Recognize approaches that pharmacists can employ to enhance patient adherence to gout treatment.
History
The prevalence of gout has doubled in the past 20 years in the United States, with evidence suggesting that elevated uric acid levels may contribute to the risk of cardiovascular disease and other comorbidities. However, the management of gout is suboptimal, with many patients not reaching the target serum uric acid goal. Pharmacists can play a key role in improving gout management and reducing associated cardiovascular risk.
Key Points
- The prevalence of gout has doubled in the past 20 years in the United States, with approximately 39% of US adults affected.
- Gout is more common in men than in women, but its occurrence in women increases after menopause.
- Gout is associated with an increased risk of cardiovascular disease, and patients who fail to reach the recommended serum uric acid goal have a higher mortality rate.
- Clinical inertia often occurs due to providers’ lack of familiarity with treatment guidelines for gout, resulting in suboptimal management practices.
Clinical presentation
- Acute gouty arthritis occurs when monosodium urate crystals exceed their solubility limit in a joint (SUA level of >6.8 mg/dL).
- This triggers an immune response, leading to inflammation and the formation of tophi, visible nodules containing the crystals.
- Gout is characterized by podagra (red, swollen first metatarsophalangeal joint) and can also affect larger lower-limb joints, smaller joints, and distal joints.
- In elderly patients, gout may present with atypical symptoms, affecting upper limbs and having a higher chance of tophi.
Risk Factors for Gout
- Comorbidities: Metabolic syndrome, diabetes, increasing BMI, and hypertension
- Diet and Lifestyle: High-purine foods, Western diet, and DASH diet
- Medications: diuretics (duration of therapy and increasing dose), other antihypertensives, low-dose aspirin
- Age-Related Factors: increased adiposity, beer and protein intake, suboptimal kidney function, smoking, hypertension

Diagnosis
Gout is typically diagnosed based on clinical presentation, which includes the rapid development of monoarticular arthritis with associated swelling and redness. While aspiration of joint fluid and identification of urate crystals are required for a confirmatory diagnosis, this is rarely done in practice. For more information, you can refer to the American College of Rheumatology (ACR)–European League Against Rheumatism (EULAR) diagnostic criteria available at this link.
Treatment guidelines
- Initiation of ULT:
- ACP guidelines: Least aggressive, does not recommend ULT after first attack or in patients with infrequent attacks.
- ACR guidelines: Recommend ULT in patients with tophi, two or more flares per year, stage 2 or higher CKD, and history of urolithiasis.
- EULAR guidelines: Recommend consideration of ULT initiation from the first attack, based on the impact of hyperuricemia on CV health.
- Agent Preference for ULT:
- ACP guidelines: No preference given.
- ACR guidelines: Recommend xanthine oxidase inhibitors (XOI) as first-line ULT, with equal consideration to allopurinol and febuxostat.
- EULAR guidelines: Recommend allopurinol due to affordability.
- Treat-to-Target Approach:
- ACR and EULAR guidelines: Recommend treating to a target SUA level of <6 mg/dL and allow for a more aggressive target of <5 mg/dL in patients with more severe disease.
- ACP guidelines: Do not recommend a treat-to-target approach or ULT initiation due to insufficient evidence of improved outcomes or adherence.
Acronyms:
- ULT: Urate-lowering therapy
- ACP: American College of Physicians
- EULAR: European League Against Rheumatism
- ACR: American College of Rheumatology
- CKD: Chronic Kidney Disease
- SUA: Serum Uric Acid
- CV: Cardiovascular
Treatment options
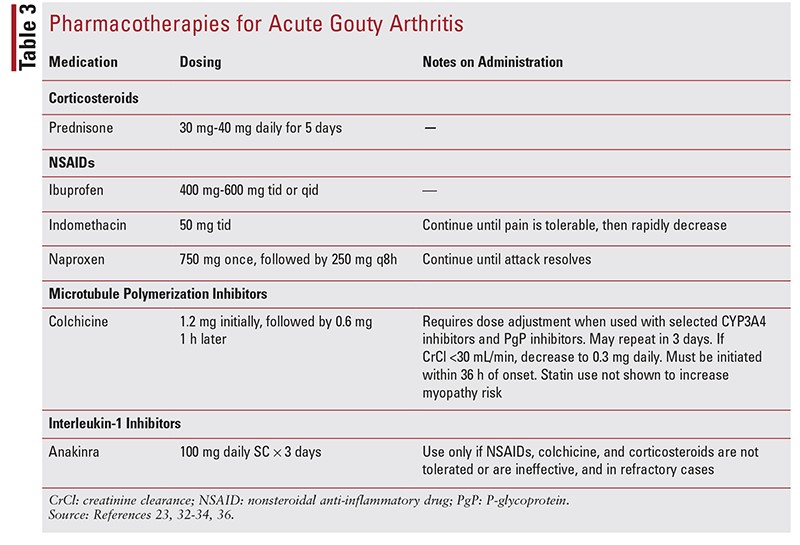
- Treatment options for acute gouty arthritis include NSAIDs, corticosteroids, colchicine, and IL-1 inhibitors. The ACR and EULAR guidelines do not have a treatment preference, but the ACP guidelines recommend corticosteroids as first-line treatment.
- Recent evidence has shown corticosteroids to be equally effective as NSAIDs for pain resolution. Colchicine is less favored due to cost increases and side effects.
- NSAIDs are recommended for patients under 60 without major comorbidities. Combination therapy of first-line treatments is allowed, except for corticosteroids and NSAIDs together due to concerns about GI toxicity.
- These treatment options are also recommended when ULT is initiated to prevent gouty arthritis exacerbations or recurrence.
- Anti-inflammatory prophylaxis with ULT may include NSAIDs, colchicine, and low-dose corticosteroids.
- If tophi are present, prophylaxis should continue for 6 months after reaching the SUA goal; if no tophi are present, prophylaxis should continue for 3 months after reaching the SUA goal.
ULT options
- FDA warned about increased risk of heart-related death with febuxostat compared to allopurinol in 2017.
- Febuxostat use is now recommended for patients who have failed to tolerate allopurinol.
- Allopurinol is generally considered the first-line treatment for gout, with febuxostat reserved for patients who are hypersensitive or intolerant to allopurinol.
- Emerging evidence suggests that allopurinol may protect against renal disease better than febuxostat.
- Lesinurad has been discontinued due to efficacy and safety concerns.
ULT pearls
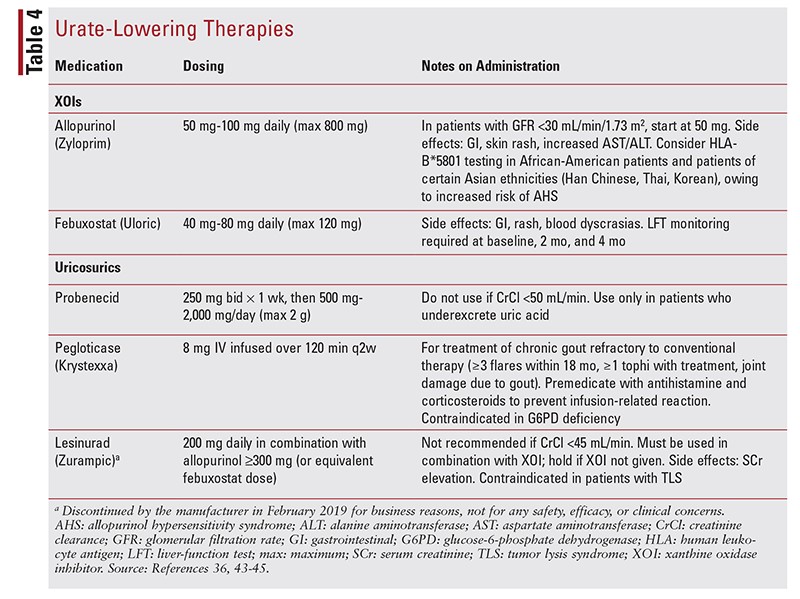
- Allopurinol and febuxostat should be initiated at a low dosage and titrated every 2 to 5 weeks until the SUA level is <6 mg/dL.
- Allopurinol dosage ranges from 100 mg to 800 mg/day, with higher doses sometimes needed for tophaceous gout.
- Febuxostat dosage is typically 80 mg/day.
- Lesinurad should be used in combination with allopurinol 300 mg daily or febuxostat 80 mg daily.
- Verinurad, another medication in this class, is currently in development, and similar renal dosing is expected.
- Fenofibrate can lower uric acid levels as much as 20% to 30% when used in combination with XOI therapy.
- Losartan can reduce uric acid levels, particularly in patients with concomitant diabetes and/or hypertension who qualify for ACE inhibitor therapy or angiotensin receptor blocker therapy.
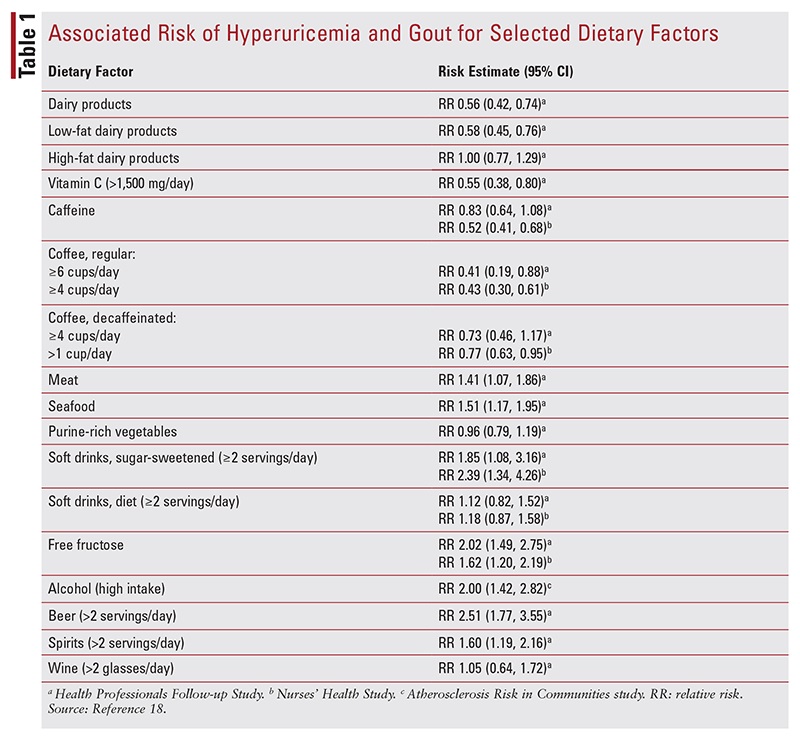
Gout and diet
Summary
Patients often seek natural therapies for rheumatologic disorders, including cherry extract, vitamin C, celery seed, and turmeric. However, there is inadequate evidence for the safety and effectiveness of cherry extract, while vitamin C has data supporting its effectiveness for preventing acute flares. Celery seed has been used for acute pain treatment during a flare, but there is no recommended dosage, and turmeric also lacks efficacy data.
Key Points
- Cherry extract should not be recommended due to inadequate evidence of safety and effectiveness
- Vitamin C has data supporting its effectiveness for preventing acute flares at dosages of 500 mg/day or more
- Celery seed has been used for acute pain treatment during a flare, but no recommended dosage or data supporting its effectiveness
- Turmeric lacks efficacy data, but a dosage of 1,000 mg/day has been found to be effective for other arthritic disorders
FAQ
Q: What is hyperuricemia and how is it related to gout?
A: Hyperuricemia is a condition characterized by an elevated level of uric acid in the blood. When the body produces an excessive amount of uric acid or is unable to expel enough of it through the kidneys, hyperuricemia may occur. High levels of uric acid can lead to the formation of uric acid crystals in the body. These crystals can accumulate in the joints causing an acute gout attack; thus, hyperuricemia is associated with the development of gout.
Q: What are the symptoms of an acute gout flare?
A: An acute gout flare, also known as a gouty arthritis attack, often occurs suddenly. It is typically characterized by severe pain, swelling, redness, and warmth in the affected joint, often the big toe. Gout symptoms can be severe and they usually peak within 24 to 48 hours.
Q: What contributes to the development of chronic gout?
A: Chronic gout occurs when a patient experiences repeated gout flares over a long period of time. This can be caused by prolonged hyperuricemia as the uric acid crystals deposit in the joints leading to permanent joint damage. Other factors include inadequate treatment of hyperuricemia and lifestyle factors like diet and alcohol consumption.
Q: Which factors can cause hyperuricemia?
A: Some causes of hyperuricemia include genetics, kidney disease, certain medications, obesity, excessive alcohol consumption, and diets high in purines, a substance that breaks down into uric acid.
Q: How is hyperuricemia diagnosed?
A: Hyperuricemia is typically diagnosed through a blood test to measure the serum uric acid level. However, high levels of uric acid alone are not enough for a diagnosis of gout. Other factors and clinical practice guidelines and consensus statements are considered. A joint fluid test to look for uric acid crystals may also be required.
Q: What is the prevalence of hyperuricemia and gout?
A: Gout is a common condition, and hyperuricemia is even more prevalent. The prevalence of hyperuricemia varies by region and demographics, but it has been increasing globally, likely due to changes in diet, lifestyle, and an increase in conditions associated with hyperuricemia, like metabolic syndrome and kidney disease.
Q: What treatment options are available for hyperuricemia and acute gout attack?
A: Acute gout attacks are usually treated with anti-inflammatory medications to alleviate pain and swelling. For the management of hyperuricemia, treatments aim at reducing serum uric acid levels. This can be achieved with lifestyle modifications and medications that reduce uric acid production or increase its excretion. A review of clinical practice guidelines may be helpful.
Q: How are patients with asymptomatic hyperuricemia managed?
A: Asymptomatic hyperuricemia refers to high levels of uric acid without symptoms of gout. Presently, rheumatology guidelines for the management of such cases prefer lifestyle modifications over medical treatment, unless there are other risk factors present.
Q: What is chronic tophaceous gout?
A: Chronic tophaceous gout is a severe and advanced stage of gout. It occurs when large deposits of uric acid crystals (tophi) accumulate in the joints and can cause deformity and damage to the joints. This stage usually occurs after many years of untreated or inadequately treated hyperuricemia and gout.
Q: What drugs are available for the treatment of hyperuricemia?
A: Several drugs are available to manage hyperuricemia. These include uric acid lowering drugs (like allopurinol and febuxostat), and uricosuric drugs which increase uric acid excretion (like probenecid). It is important that treatment decisions are individualized considering the patient’s overall health, associated diseases, and the risk of ongoing gout flares.
REFERENCES
- Elfishawi MM, Zleik N, Kvrgic Z, et al. The rising incidence of gout and the increasing burden of comorbidities: a population-based study over 20 years. J Rheumatol. 2018;45:574-579.
- Chen-Xu M, Yokose C, Rai SK, et Contemporary prevalence of gout and hyperuricemia in the United States and decadal trends: the National Health and Nutrition Examination Survey, 2007-2016. Arthritis Rheumatol. 2019;71:991-999.
- Hainer BL, Matheson E, Wilkes Diagnosis, treatment, and prevention of gout. Am Fam Physician. 2014;90:831-836.
- Vargas-Santos AB, Neogi Management of gout and hyperuricemia in CKD. Am J Kidney Dis. 2017;70:422-439.
- Burke BT, Köttgen A, Law A, et al. Gout in older adults: the Atherosclerosis Risk in Communities study. J Gerontol A Biol Sci Med Sci. 2016;71:536-542.
- Feig DI, Kang DH, Johnson Uric acid and cardiovascular risk. N Engl J Med. 2008;359:1811-1821.
- Culleton BF, Larson MG, Kannel WB, Levy D. Serum uric acid and risk for cardiovascular disease and death: the Framingham Heart Study. Ann Intern Med. 1999;131:7-13.
- Pérez Ruiz FP, Richette P, Stack AG, et Failure to reach uric acid target of <0.36 mmol/L in hyperuricaemia of gout is associated with elevated total and cardiovascular mortality. RMD Open. 2019;5:e001015.
- Ioachimescu AG, Brennan DM, Hoar BM, et Serum uric acid is an independent predictor of all-cause mortality in patients at high risk of cardiovascular disease: a preventive cardiology information system (PreCIS) database cohort study. Arthritis Rheum. 2008;58:623-630.
- Harrold LR, Mazor KM, Negron A, et al. Primary care providers’ knowledge, beliefs and treatment practices for gout: results of a physician questionnaire. Rheumatology (Oxford). 2013;52:16231629.
- Maravic M, Hincapie N, Pilet S, et Persistent clinical inertia in gout in 2014: an observational French longitudinal patient database study. Joint Bone Spine. 2018;85:311-315.
- Neogi Clinical practice. Gout. N Engl J Med. 2011;364:443452.
- Terkeltaub R. Update on gout: new therapeutic strategies and options. Nat Rev Rheumatol. 2010;6:30-38.
- Gonzalez EB. An update on the pathology and clinical management of gouty arthritis. Clin Rheumatol. 2012;31:13-21.
- Haines A, Bolt J, Dumont Z, Semchuk Pharmacists’ assessment and management of acute and chronic gout. Can Pharm J (Ott). 2018;151:107-113.
- Camerota A, Mastroiacovo D, Bocale R, Desideri G. Therapeutic challenges in the management of chronic hyperuricemia and gout in the elderly. Ann Gerontol Geriatric Res. 2014;1:1012.
- Chowalloor PV, Keen HI, Inderjeeth Gout in the elderly. OA Elderly Med. 2013;1:2.
- Roddy E, Choi HK. Epidemiology of gout. Rheum Dis Clin North Am. 2014;40:155-175.
- Rai SK, Fung TT, Lu N, et al. The Dietary Approaches to Stop Hypertension (DASH) diet, Western diet, and risk of gout in men: prospective cohort study. 2017;357:j1794.
- Holland R, McGill Comprehensive dietary education in treated gout patients does not further improve serum urate. Intern Med J. 2015;45:189-194.
- Choi HK. Diet, alcohol, and gout: how do we advise patients given recent developments? Curr Rheumatol Rep. 2005;7:220-226.
- Hunter DJ, York M, Chaisson CE, et al. Recent diuretic use and the risk of recurrent gout attacks: the online case-crossover gout study. J Rheumatol. 2006;33:1341-1345.
- Wilson L, Saseen JJ. Gouty arthritis: a review of acute management and prevention. 2016;36:906-922.
- Juraschek SP, Appel LJ, Miller ER III. Metoprolol increases uric acid and risk of gout in African Americans with chronic kidney disease attributed to hypertension. Am J Hypertens. 2017;30:871-875.
- Vezmar Kovacevic S, Simisic M, Stojkov Rudinski S, et al. Potentially inappropriate prescribing in older primary care patients. PLoS One. 2014;9:e95536.
- Zhang Y, Neogi T, Chen C, et al. Low-dose aspirin use and recurrent gout Ann Rheum Dis. 2014;73:385-390.
- Qaseem A, Harris RP, Forciea MA. Management of acute and recurrent gout: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166:58-68
- Khanna D, Fitzgerald JD, Khanna PP, et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res (Hoboken). 2012;64(10):1431-1446.
- Khanna D, Khanna PP, Fitzgerald JD, et al. 2012 American College of Rheumatology guidelines for management of gout. Part 2: therapy and anti-inflammatory prophylaxis of acute gouty arthritis. Arthritis Care Res (Hoboken). 2012;64:1447-1461.
- Richette P, Doherty M, Pascual E, et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis. 2017;76:29-42.
- Doherty M, Jenkins W, Richardson H, et al. Efficacy and cost-effectiveness of nurse-led care involving education and engagement of patients and a treat-to-target urate-lowering strategy versus usual care for gout: a randomised controlled trial. 2018;392:1403-1412.
- Prasad S, Ewigman B. Acute gout: oral steroids work as well as NSAIDs. J Fam Pract. 2008;57:655-657.
- Wilbur K, Makowsky M. Colchicine myotoxicity: case reports and literature review. Pharmacotherapy. 2004;24:1784-1792.
- Kwon OC, Hong S, Ghang B, et al. Risk of colchicine-associated myopathy in gout: influence of concomitant use of statin. Am J Med. 2017;130:583-587.
- American College of Rheumatology. Gout. rheumatology.org/Practice-Quality/Clinical-Support/Clinical-Practice-Guidelines/Gout. Accessed January 7, 2020.
- Duzallo and Zurampic. duzallohcp.com. Accessed February 14, 2020.
- Janssens HJ, Janssen M, van de Lisdonk EH, et al. Use of oral prednisolone or naproxen for the treatment of gout arthritis: a double-blind, randomised equivalence trial. 2008;371:1854-1860.
- Abhishek A, Valdes AM, Jenkins W, et al. Triggers of acute attacks of gout, does age of onset matter? A primary care based cross-sectional PLoS One. 2017;12:e0186096.
- Celebrex (celecoxib) package insert. New York, NY: Pfizer Inc; May 2019.
- FDA adds Boxed Warning for increased risk of death with gout medicine Uloric (febuxostat). www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-increased-risk-death-gout-medicine-uloricfebuxostat. Accessed January 15, 2020.
- White WB, Saag KG, Becker MA, et al. Cardiovascular safety of febuxostat or allopurinol in patients with gout. N Engl J Med. 2018;378:1200-1210.
- FDA to evaluate increased risk of heart-related death and death from all causes with the gout medication febuxostat (Uloric). www.fda. gov/media/108760/download. Accessed January 15, 2020.
- Gillen M, Valdez S, Zhou D, et al. Effects of renal function on pharmacokinetics and pharmacodynamics of lesinurad in adult volunteers. Drug Des Devel Ther. 2016;10:3555-3562.
- Gupta A, Sharma PK, Misra AK, Singh S. Lesinurad: a significant advancement or just another addition to existing therapies of gout? J Pharmacol Pharmacother. 2016;7:155-158.
- Koide H, Hira D, Tsujimoto M, et al. Previous dosage of allopurinol is a strong determinant of febuxostat efficacy. Biol Pharm Bull. 2017;40:681686.
- Cutolo M, Cimmino MA, Perez-Ruiz Potency on lowering serum uric acid in gout patients: a pooled analysis of registrative studies comparing febuxostat vs. allopurinol. Eur Rev Med Pharmacol Sci. 2017;21:41864195.
- Becker MA, Schumacher HR, Espinoza LR, et al. The urate-lowering efficacy and safety of febuxostat in the treatment of the hyperuricemia of gout: the CONFIRMS trial. Arthritis Res Ther. 2010;12:R63.
- Stamp LK, Chapman Urate-lowering therapy: current options and future prospects for elderly patients with gout. Drugs Aging. 2014;31:777786.
- Singh JA, Cleveland JD. Gout and the risk of age-related macular degeneration in the PLoS One. 2018;13:e0199562.
- Singh JA, Yu Are allopurinol dose and duration of use nephroprotective in the elderly? A Medicare claims study of allopurinol use and incident renal failure. Ann Rheum Dis. 2017;76:133-139.
- Levy G, Shi JM, Cheetham TC, Rashid N. Urate-lowering therapy in moderate to severe chronic kidney disease. Perm J. 2018;22:17-142.
- Sircar D, Chatterjee S, Waikhom R, et al. Efficacy of febuxostat for slowing the GFR decline in patients with CKD and asymptomatic hyperuricemia: a 6-month, double-blind, randomized, placebo-controlled trial. Am J Kidney Dis. 2015;66:945-950.
- Graham GG, Kannangara DR, Stocker SL, et al. Understanding the dose-response relationship of allopurinol: predicting the optimal Br J Clin Pharmacol. 2013;76:932-938.
- Zurampic (lesinurad) package insert. Cambridge, MA: Ironwood Pharmaceuticals, Inc; January
- Smith WB, Hall J, Berg JK, et al. Effect of renal impairment on the pharmacokinetics and pharmacodynamics of verinurad, a selective uric acid reabsorption inhibitor. Clin Drug Investig. 2018;38:703-713.
- Jung JY, Choi Y, Suh CH, et al. Effect of fenofibrate on uric acid level in patients with Sci Rep. 2018;8:16767.
- Wolff ML, Cruz JL, Vanderman AJ, Brown JN. The effect of angiotensin II receptor blockers on hyperuricemia. Ther Adv Chronic Dis. 2015;6:339-346.
- Singh JA, Shah N, Edwards NL. A cross-sectional internet-based patient survey of the management strategies for gout. BMC Complement Altern Med. 2016;16:90.
- Natural Medicines (comprehensive database). https://naturalmedicines. therapeuticresearch.com. Accessed February 13,
- Daily JW, Yang M, Park S. Efficacy of turmeric extracts and curcumin for alleviating the symptoms of joint arthritis: a systematic review and meta-analysis of randomized clinical J Med Food. 2016;19:717-729.
- Aung T, Myung G, FitzGerald JD. Treatment approaches and adherence to urate-lowering therapy for patients with gout. Patient Prefer Adherence. 2017;11:795-800.
- Morlock R, Chevalier P, Horne L, et Disease control, health resource use, healthcare costs, and predictors in gout patients in the United States, the United Kingdom, Germany, and France: a retrospective analysis. Rheumatol Ther. 2016;3:53-75.
- Briesacher BA, Andrade SE, Fouayzi H, Chan KA. Comparison of drug adherence rates among patients with seven different medical conditions. Pharmacotherapy. 2008;28:437-443.
- Nassar-Ghodsi N, Harrold LR. Overcoming adherence issues and other barriers to optimal care in gout. Curr Opin Rheumatol. 2015;27:134-138.
- Singh JA. Challenges faced by patients in gout treatment: a qualitative study. J Clin Rheumatol. 2014;20:172-174.
- Goldfien RD, Ng MS, Yip G, et al. Effectiveness of a pharmacist-based gout care management programme in a large integrated health plan: results from a pilot study. BMJ Open. 2014;4:e003627.